DEF 14A: Definitive proxy statements
Published on April 22, 2026
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
Proxy Statement Pursuant to Section 14(a) of the
Securities Exchange Act of 1934
(AMENDMENT NO. )
Filed by the Registrant ☒ Filed by a Party other than the Registrant ☐
Check the appropriate box:
☐ Preliminary Proxy Statement
☐ Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2))
☒ Definitive Proxy Statement
☐ Definitive Additional Materials
☐ Soliciting Material Pursuant to §240.14a-12
(Name of Registrant as Specified In Its Charter)
N/A
(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
Payment of Filing Fee (Check the appropriate box):
☒ No fee required.
☐ Fee paid previously with preliminary materials.
☐ Fee computed on table in exhibit required by Item 25(b) per Exchange Act Rules 14a-6(i)(1) and 0-11.
SUTRO BIOPHARMA, INC.
111 Oyster Point Boulevard
South San Francisco, California, 94080
NOTICE OF ANNUAL MEETING OF SHAREHOLDERS
To Be Held June 5, 2026
To Our Stockholders:
NOTICE IS HEREBY GIVEN that the 2026 Annual Meeting of Shareholders of Sutro Biopharma, Inc. will be held via a virtual meeting. You will be able to participate in the 2026 Annual Meeting and vote during the 2026 Annual Meeting via live webcast by visiting www.virtualshareholdermeeting.com/STRO2026 on Friday, June 5, 2026 at 8:00 AM Pacific Time. We believe that a virtual stockholder meeting provides greater access to those who may want to attend, and therefore we have chosen this over an in-person meeting. It is important that you retain a copy of the control number found on the proxy card or voting instruction form, or included in the e-mail to you if you received the proxy materials by e-mail, as such number will be required in order for stockholders to gain access to the virtual meeting.
We are holding the meeting for the following purposes, which are more fully described in the accompanying proxy statement:
1.To elect three Class II directors, each to serve three-year terms through the third annual meeting of shareholders following this meeting and until a successor has been elected and qualified or until earlier resignation or removal.
2.To ratify the appointment of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026.
3.To approve, on a non-binding advisory basis, the compensation of our named executive officers as disclosed in the proxy statement for the 2026 Annual Meeting.
In addition, stockholders may be asked to consider and vote upon such other business as may properly come before the meeting or any adjournment or postponement thereof.
Only stockholders of record at the close of business on April 7, 2026 are entitled to receive notice of, and to vote at, the meeting and any adjournments thereof. This Notice of Internet Availability of Proxy Materials (“Notice of Internet Availability”) and the accompanying proxy statement are being mailed out to stockholders as of the record date beginning on or about April 22, 2026.
For ten days prior to the meeting, a complete list of the stockholders entitled to vote at the meeting will be available upon request by any stockholder for any purpose relating to the meeting. Stockholders can request the list of stockholders through our investor relations website at https://ir.sutrobio.com/contact-ir.
Your vote as a Sutro Biopharma, Inc. stockholder is very important. Each share of common stock that you own represents one vote.
For questions regarding your stock ownership, you may contact our Investor Relations group at IR@sutrobio.com or, if you are a registered holder, our transfer agent, Equiniti Trust Company, LLC by email through their website at https://www.equiniti.com/us or by phone at (800) 937-5449. Whether or not you expect to attend the meeting, we encourage you to read the proxy statement and vote through the internet or by telephone, or to request, sign and return your proxy card as soon as possible, so that your shares may be represented at the meeting. For specific instructions on how to vote your shares, please refer to the section entitled “General Proxy Information” in the proxy statement.
By Order of the Board of Directors, |
|
Jane Chung |
Chief Executive Officer and Director |
South San Francisco, California
April 22, 2026
Important Notice Regarding the Availability of Proxy Materials for the virtual Annual Meeting of Shareholders to be held on June 5, 2026: the Proxy Statement and our 2025 Annual Report on Form 10-K are available at http://ir.sutrobio.com. You will need the control number included on your proxy card or voting instruction form, or included in the e-mail to you if you received the proxy materials by e-mail, as such number will be required in order for stockholders to gain access to the virtual meeting.
SUTRO BIOPHARMA, INC.
PROXY STATEMENT FOR 2026 ANNUAL MEETING OF SHAREHOLDERS
TABLE OF CONTENTS
|
Page |
1 |
|
|
|
1 |
|
|
|
1 |
|
|
|
2 |
|
|
|
5 |
|
|
|
12 |
|
|
|
PROPOSAL NO. 2 RATIFICATION OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM |
18 |
|
|
19 |
|
|
|
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT |
20 |
|
|
24 |
|
|
|
40 |
|
|
|
43 |
|
|
|
44 |
|
|
|
45 |
|
|
|
45 |
|
|
|
46 |
SUTRO BIOPHARMA, INC.
111 Oyster Point Boulevard
South San Francisco, California, 94080
PROXY STATEMENT FOR THE 2026 ANNUAL MEETING OF SHAREHOLDERS
April 22, 2026
INFORMATION ABOUT SOLICITATION AND VOTING
The accompanying proxy is solicited on behalf of the Board of Directors of Sutro Biopharma, Inc. (Sutro Biopharma or the Company) for use at Sutro Biopharma’s 2026 Annual Meeting of Shareholders (Annual Meeting) to be held via a virtual meeting. You will be able to participate in the Annual Meeting and vote during the Annual Meeting via live webcast by visiting www.virtualshareholdermeeting.com/STRO2026 on Friday, June 5, 2026 at 8:00 AM Pacific Time and any adjournment or postponement thereof. We believe that a virtual stockholder meeting provides greater access to those who may want to attend, and therefore we have chosen this format over an in-person meeting. This approach also lowers costs and aligns with our broader sustainability goals. You will need the control number included on your proxy card or voting instruction form, or included in the e-mail to you if you received the proxy materials by e-mail, as such number will be required in order for stockholders to gain access to the virtual meeting. We are making this proxy statement, the accompanying form of proxy and our Annual Report on Form 10‑K for the year ended December 31, 2025 first available to stockholders on or about April 22, 2026. An electronic copy of this proxy statement and Annual Report on Form 10‑K are available at http://ir.sutrobio.com.
Except as otherwise indicated, all share and per share information in this Proxy Statement is presented including the effect of the reverse stock split of the Company’s outstanding common stock at a ratio of one-for-ten shares, which was effected on December 2, 2025.
INTERNET AVAILABILITY OF PROXY MATERIALS
Under rules adopted by the Securities and Exchange Commission (the “SEC”), we are furnishing proxy materials to our stockholders primarily via the Internet, instead of mailing printed copies to each stockholder. On or about April 22, 2026, we expect to send to our stockholders a Notice of Internet Availability containing instructions on how to access our proxy materials, including our proxy statement and our Annual Report on Form 10-K. The Notice of Internet Availability also provides instructions on how to vote and includes instructions on how to receive paper copies of the proxy materials by mail, or an electronic copy of the proxy materials by email.
This process is designed to reduce our environmental impact and lower the costs of printing and distributing our proxy materials while providing our stockholders timely access to this important information. If you would prefer to receive printed proxy materials, please follow the instructions included in the Notice of Internet Availability.
GENERAL INFORMATION ABOUT THE MEETING
Purpose of the Meeting
At the meeting, stockholders will act upon the proposals described in this proxy statement. In addition, we will consider any other matters that are properly presented for a vote at the meeting. We are not aware of any other matters to be submitted for consideration at the meeting. If any other matters are properly presented for a vote at the meeting, the persons named in the proxy, who are officers of the company, have the authority in their discretion to vote the shares represented by the proxy.
Record Date; Quorum
Only holders of record of common stock at the close of business on April 7, 2026, the record date, will be entitled to vote at the meeting. At the close of business on April 7, 2026, 16,567,887 shares of common stock were outstanding and entitled to vote.
1
The holders of a majority of the voting power of the shares of stock entitled to vote at the meeting as of the record date must be present or represented by proxy at the meeting in order to hold the meeting and conduct business. This presence is called a quorum. Your shares are counted as present at the meeting if you are present and vote online at the virtual meeting or if you have properly submitted a proxy.
GENERAL PROXY INFORMATION
Voting Rights; Required Vote
Each holder of shares of common stock is entitled to one vote for each share of common stock held as of the close of business on April 7, 2026, the record date. You may vote all shares owned by you at such date, including (1) shares held directly in your name as the stockholder of record and (2) shares held for you as the beneficial owner in street name through a broker, bank, trustee or other nominee. Dissenters’ rights are not applicable to any of the matters being voted on.
Stockholder of Record: Shares Registered in Your Name. If on April 7, 2026, your shares were registered directly in your name with our transfer agent, Equiniti Trust Company LLC, then you are considered the stockholder of record with respect to those shares. As a stockholder of record, you may vote at the meeting, or vote in advance through the internet or by telephone, or if you request to receive paper proxy materials by mail, by filling out and returning the proxy card.
Beneficial Owner: Shares Registered in the Name of a Broker or Nominee. If on April 7, 2026, your shares were held in an account with a brokerage firm, bank or other nominee, then you are the beneficial owner of the shares held in street name. As a beneficial owner, you have the right to direct your broker on how to vote the shares held in your account, and your broker has enclosed or provided voting instructions for you to use in directing it on how to vote your shares. Because the brokerage firm, bank or other nominee that holds your shares is the stockholder of record, if you wish to attend the meeting and vote your shares, you must obtain a valid proxy from the firm that holds your shares giving you the right to vote the shares at the meeting.
Each director will be elected by a plurality of the votes cast by the holders of the shares present in person or represented by proxy at the meeting and entitled to vote on the election of directors. This means that the three individuals nominated for election to the Board of Directors at the meeting receiving the highest number of “FOR” votes will be elected. You may either vote “FOR” one, two or all three of the nominees or “WITHHOLD” your vote with respect to one, two or all three of the nominees. You may not cumulate votes in the election of directors. Approval of the ratification of the appointment of our independent registered public accounting firm requires the affirmative vote of the holders of a majority of the voting power of the shares of stock entitled to vote on such matter that are present in person or represented by proxy at the meeting and are voted for or against the matter. Approval, on a non-binding advisory basis, of the compensation of our named executive officers requires the affirmative vote of the holders of a majority of the voting power of the shares of stock entitled to vote on such matter that are present in person or represented by proxy at the meeting and are voted for or against the matter.
A proxy submitted by a stockholder may indicate that the shares represented by the proxy are not being voted (stockholder withholding) with respect to a particular matter or the stockholder may abstain from voting on the matter (abstention). In addition, a broker may not be permitted to vote on shares held in street name on a particular matter in the absence of instructions from the beneficial owner of the stock (broker non-vote). The shares subject to a proxy which are not being voted on a particular matter because of either stockholder withholding or broker non-votes will count for purposes of determining the presence of a quorum, but are not treated as votes cast and, therefore, will have no effect on the outcome of any of the proposals. Additionally, abstentions and votes withheld will count for purposes of determining the presence of a quorum, but are voted neither “for” nor “against” a matter, and, therefore, will have no effect on the outcome of any of the proposals.
2
Recommendations of the Board of Directors on Each of the Proposals Scheduled to be Voted on at the Meeting
The Board of Directors recommends that you vote FOR the election of each of the Class II directors named in this proxy statement (Proposal 1); FOR the ratification of the appointment of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026 (Proposal 2); and FOR the approval, on a non-binding advisory basis, of the compensation of our named executive officers, as disclosed in this Proxy Statement (Proposal 3).
None of the directors or executive officers has any substantial interest in any matter to be acted upon, other than elections to office with respect to the directors nominated in Proposal 1.
Voting Instructions; Voting of Proxies
If you are a stockholder of record, you may:
Votes submitted through the internet or by telephone must be received by 11:59 p.m., Eastern Time, on June 4, 2026. Submitting your proxy, whether by telephone, through the internet or by mail if you requested or received a paper proxy card, will not affect your right to vote online should you decide to attend the virtual meeting. If you are not the stockholder of record, please refer to the voting instructions provided by your nominee to direct how to vote your shares. For Proposal 1, you may either vote “FOR” all of the nominees to the Board of Directors, or you may withhold your vote from any nominee you specify. For Proposal 2, you may vote “FOR” or “AGAINST” or “ABSTAIN” from voting. For Proposal 3, you may vote “FOR” or “AGAINST” or “ABSTAIN” from voting. Your vote is important. Whether or not you plan to attend the meeting, we urge you to vote by proxy to ensure that your vote is counted.
All proxies will be voted in accordance with the instructions specified on the proxy card. If you sign a physical proxy card and return it without instructions as to how your shares should be voted on a particular proposal at the meeting, your shares will be voted in accordance with the recommendations of our Board of Directors stated above.
If your shares are held in an account with a brokerage firm, bank or other nominee, then you are deemed to be the beneficial owner of your shares and the broker that actually holds the shares for you is the record holder and is required to vote the shares it holds on your behalf according to your instructions. The proxy materials, as well as voting and revocation instructions, should have been forwarded to you by the bank, broker or other nominee that holds your shares. In order to vote your shares, you will need to follow the instructions that your bank, broker or other nominee provides you. The voting deadlines and availability of telephone and Internet voting for beneficial owners of shares held in street name will depend on the voting processes of the bank, broker or other nominee that holds your shares. Therefore, we urge you to carefully review and follow the voting instruction card and any other materials that you receive from that organization.
If you received a Notice of Internet Availability, please follow the instructions included on the notice on how to access and vote your proxy card. If you do not vote and you hold your shares in street name, and your broker does not have discretionary power to vote your shares, your shares may constitute “broker non-votes” (as described above) and will not be counted in determining the number of shares necessary for approval of the proposals.
3
However, shares that constitute broker non-votes will be counted for the purpose of establishing a quorum for the meeting.
If you receive more than one proxy card or Notice of Internet Availability, your shares are registered in more than one name or are registered in different accounts. To make certain all of your shares are voted, please follow the instructions included on the Notice of Internet Availability on how to access and vote each proxy card. If you requested or received paper proxy materials by mail, please complete, sign, date and return each proxy card to ensure that all of your shares are voted.
Expenses of Soliciting Proxies
We will pay the expenses associated with soliciting proxies. Following the original distribution and mailing of the solicitation materials, we or our agents may solicit proxies by mail, email, telephone, facsimile, by other similar means, or in person. Our directors, officers and other employees, without additional compensation, may solicit proxies personally or in writing, by telephone, email or otherwise. Following the original distribution and mailing of the solicitation materials, we will request brokers, custodians, nominees and other record holders to forward copies of those materials to persons for whom they hold shares and to request authority for the exercise of proxies. In such cases, we, upon the request of the record holders, will reimburse such holders for their reasonable expenses. If you choose to access the proxy materials and/or vote through the internet, you are responsible for any internet access charges you may incur.
Revocability of Proxies
A stockholder of record who has given a proxy may revoke it at any time before the closing of the polls by the inspector of elections at the meeting by:
Please note, however, that if your shares are held of record by a brokerage firm, bank or other nominee, and you wish to revoke a proxy, you must contact that firm to revoke or change any prior voting instructions.
Electronic Access to the Proxy Materials
The Notice of Internet Availability will provide you with instructions regarding how to:
Choosing to receive your future proxy materials by email will reduce the impact of our annual meetings of stockholders on the environment and lower the costs of printing and distributing our proxy materials. If you choose to receive future proxy materials by email, you will receive an email next year with instructions containing a link to those materials and a link to the proxy voting site. Your election to receive proxy materials by email will remain in effect until you terminate it.
4
Voting Results
Voting results will be tabulated and certified by the inspector of elections appointed for the meeting. The final results will be tallied by the inspector of elections and filed with the SEC in a Current Report on Form 8-K within four business days of the meeting.
CORPORATE GOVERNANCE STANDARDS AND DIRECTOR INDEPENDENCE
We are committed to good corporate governance practices. These practices provide an important framework within which our Board of Directors and management pursue our strategic objectives for the benefit of our stockholders.
Corporate Governance Guidelines
Our Board of Directors has adopted Corporate Governance Guidelines that set forth expectations for directors, director independence standards, Board Committee structure and functions, and other policies for the governance of the company. Our Corporate Governance Guidelines are available without charge on the investor relations section of our website at https://ir.sutrobio.com/corporate-governance/governance-documents.
Board Composition and Leadership Structure
Our Nominating and Governance Committee periodically considers the leadership structure of our Board of Directors and makes such recommendations to our Board of Directors as our Nominating and Governance Committee deems appropriate. The positions of Chief Executive Officer and Chair of our Board of Directors are currently held by two different individuals (Jane Chung and Connie Matsui, respectively). This structure allows our Chief Executive Officer to focus on our day-to-day business while our Chair leads our Board of Directors in its fundamental role of providing advice to, and independent oversight of, management. Ms. Chung’s significant managerial experience working with and serving in various executive positions in life sciences companies makes her well-suited for this day-to-day operational role, while Ms. Matsui’s extensive experience advising life sciences companies with respect to corporate strategy, product development, sales and marketing and operations allows her to perform an oversight function separate from management. Our Board of Directors believes such separation is appropriate, as it enhances the accountability of the Chief Executive Officer to the Board of Directors and strengthens the independence of the Board of Directors from management. Any changes to the leadership structure of our Board of Directors, if made, will be promptly disclosed on the investor relations section of our website and in our proxy materials. Our Board of Directors, in its sole discretion, may seek input from our stockholders on the leadership structure of the Board of Directors.
Board’s Role in Risk Oversight
Our Board of Directors believes that open communication between management and the Board of Directors is essential for effective risk management and oversight. Our Board of Directors meets with our Chief Executive Officer and other members of the senior management team at regular quarterly Board of Director meetings, and at ad hoc meetings when deemed appropriate, where, among other topics, they discuss strategy and risks in the context of reports from the management team and evaluate the risks inherent in significant transactions. While our Board of Directors is ultimately responsible for risk oversight, our Board Committees assist the Board of Directors in fulfilling its oversight responsibilities in certain areas of risk. The Audit Committee assists our Board of Directors in fulfilling its oversight responsibilities with respect to risk management in the areas of internal controls over financial reporting, disclosure controls and procedures, and information security. The Compensation Committee assists our Board of Directors in assessing risks created by the incentives inherent in our compensation policies. The Nominating and Governance Committee assists our Board of Directors in fulfilling its oversight responsibilities with respect to the management of corporate, legal and regulatory risk and environmental, social and governance, or ESG, concerns. The Science and Technology Committee assists our Board of Directors in fulfilling its oversight responsibilities with respect to our research and development and platform programs.
5
Cybersecurity Risk Oversight
Our Board of Directors recognizes the critical importance of maintaining the trust and confidence of our patients, business partners and employees. Our Board of Directors is actively involved in oversight of our risk management program, and cybersecurity represents an important component of our overall approach to enterprise risk management (“ERM”). Through our ERM program, risks are identified, assessed and managed at the organization level, mission and business process level, and information system level. Our cybersecurity program, policies and procedures are fully integrated into our ERM program and are maintained in accordance with industry good standards. Our Board of Directors’ oversight of cybersecurity risk management is supported by our Audit Committee, which regularly interacts with our executive leadership, including our Chief Executive Officer, Chief Financial Officer and our General Counsel and other key officers.
We have implemented a comprehensive, cross-functional approach to identifying, preventing and mitigating cybersecurity threats and incidents, while also implementing controls and procedures that provide for the prompt escalation of certain cybersecurity incidents so that decisions regarding the public disclosure and reporting of such incidents can be made by management in a timely manner. We also maintain a comprehensive, risk-based approach to identifying and overseeing cybersecurity risks presented by third parties, including vendors, service providers and other external users of our systems, as well as the systems of third parties that could adversely impact our business in the event of a cybersecurity incident affecting those third-party systems. Further information relating to cybersecurity and information security is contained in the section titled “Cybersecurity” in our Annual Report on Form 10-K for the year ended December 31, 2025.
Director Independence
Our common stock is listed on the Nasdaq Global Market. Under the rules of the Nasdaq Stock Market, independent directors must constitute a majority of a listed company’s Board of Directors. In addition, the rules of the Nasdaq Stock Market require that, subject to specified exceptions, each member of a listed company’s Audit, Compensation and Nominating and Governance Committees must be an “independent director”. Under the rules of the Nasdaq Stock Market, a director will only qualify as an “independent director” if, in the opinion of that company’s Board of Directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. Additionally, Compensation Committee members must not have a relationship with the listed company that is material to the director’s ability to be independent from management in connection with the duties of a Compensation Committee member.
Audit Committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Securities Exchange Act of 1934, as amended (Exchange Act). In order to be considered independent for purposes of Rule 10A-3, a member of an Audit Committee of a listed company may not, other than in his or her capacity as a member of the Audit Committee, the Board of Directors or any other Board Committee: (i) accept, directly or indirectly, any consulting, advisory or other compensatory fee from the listed company or any of its subsidiaries or (ii) be an affiliated person of the listed company or any of its subsidiaries.
Our Board of Directors has undertaken a review of the independence of each director and considered whether each director has a material relationship with us that could compromise his or her ability to exercise independent judgment in carrying out his or her responsibilities. As a result of this review, our Board of Directors determined that Connie Matsui, Michael Dybbs, Sukhi Jagpal, Heidi Hunter, Joseph Lobacki, James Panek, Daniel Petree, and Jon Wigginton, representing eight of our nine incumbent directors, are “independent directors” as defined under the applicable rules and regulations of the SEC and the listing requirements and rules of the Nasdaq Stock Market. In making these determinations, our Board of Directors reviewed and discussed information provided by the directors and us with regard to each directors’ business and personal activities and relationships as they may relate to us and our management, including the beneficial ownership of our capital stock by each non-employee director and any affiliates.
Board of Directors and Committee Self-Evaluations
Our Board of Directors is committed to a robust self-evaluation process designed for continuous improvement. To achieve this, our Board of Directors conducts an annual self-evaluation for itself and its committees. As part of this
6
process, each member of the Board of Directors completes a survey to provide feedback on the processes, structure, composition and effectiveness of the Board of Directors. The survey is a detailed written questionnaire designed to help the Board of Directors assess the performance of the Board of Directors and its committees, their own individual performance and the individual performances of fellow directors. The feedback received is shared first with the Nominating and Governance Committee and then made available to the individual directors and the full Board of Directors.
Committees of Our Board of Directors
Our Board of Directors has established an Audit Committee, a Compensation Committee, a Nominating and Governance Committee and a Science and Technology Committee, each of which has the composition and responsibilities described below. Members serve on these Committees until their resignation or until otherwise determined by our Board of Directors. Each of these Committees has a written charter, copies of which are available without charge on the investor relations section of our website at https://ir.sutrobio.com/corporate-governance/governance-documents under the heading “Governance.” The Board of Directors also establishes ad hoc committees to manage particular strategic priorities from time to time, including committees to manage Business Development, Financing and Pricing, and Corporate Strategy, as appropriate.
Audit Committee
Our Audit Committee is currently composed of Mr. Jagpal, Ms. Hunter, and Mr. Lobacki. Mr. Jagpal is the Chair of our Audit Committee. The composition of our Audit Committee meets the requirements for independence under the current Nasdaq Stock Market and SEC rules and regulations. Each member of our Audit Committee is financially literate. In addition, our Board of Directors has determined that each of Mr. Jagpal and Ms. Hunter is an “Audit Committee financial expert” as defined in Item 407(d)(5)(ii) of Regulation S-K promulgated under the Securities Act. Our Audit Committee is directly responsible for, among other things:
Compensation Committee
Our Compensation Committee is composed of Mr. Lobacki, Mr. Petree, and Dr. Wigginton. Mr. Lobacki is the Chair of our Compensation Committee. The composition of our Compensation Committee meets the requirements for independence under the current Nasdaq Stock Market and SEC rules and regulations. Our Compensation Committee is responsible for, among other things:
7
The Compensation Committee has the sole authority and responsibility, subject to any approval by the Board of Directors which the Compensation Committee or legal counsel determines to be desirable or required by applicable law or the Nasdaq rules, to determine all aspects of executive compensation packages for the Chief Executive Officer and other executive officers. The Compensation Committee also makes recommendations to our Board of Directors regarding the form and amount of compensation of non-employee directors. The Compensation Committee may take into account the recommendations of the Chief Executive Officer with respect to compensation of the other executive officers, and the recommendations of the Board of Directors or any member of the Board of Directors with respect to compensation of the Chief Executive Officer and other executive officers.
The Compensation Committee engaged an independent executive compensation consulting firm, Frederic W. Cook & Co., Inc., or FW Cook, to evaluate our executive compensation and Board of Directors compensation program and practices and to provide advice and ongoing assistance on these matters for the fiscal year ended December 31, 2025. Specifically, FW Cook was engaged to:
Representatives of FW Cook met informally with the Chair of the Compensation Committee and attended the regular meetings of the Compensation Committee, including executive sessions from time to time without any members of management present. During the fiscal year ended December 31, 2025, FW Cook worked with the Compensation Committee to assist the Committee in satisfying its responsibilities and undertook no projects for management without the Committee’s prior approval. The Compensation Committee has determined that none of the work performed by FW Cook during the fiscal year ended December 31, 2025 raised any conflict of interest.
Nominating and Governance Committee
Our Nominating and Governance Committee is composed of Mr. Panek, Ms. Matsui and Mr. Petree. Mr. Panek is the Chair of our Nominating and Governance Committee. Our Nominating and Governance Committee is responsible for, among other things:
Science and Technology Committee
Our Science and Technology Committee is composed of Dr. Wigginton, Dr. Dybbs, Ms. Hunter and Mr. Panek. Dr. Wigginton is the Chair of our Science and Technology Committee. Our Science and Technology Committee is responsible for, among other things:
8
Code of Business Conduct and Ethics
Our Board of Directors has adopted a code of business conduct and ethics that applies to all of our employees, officers and directors, including our Chief Executive Officer, Chief Financial Officer and other executive and senior financial officers. We intend to disclose future amendments to certain provisions of our code of business conduct, or waivers of these provisions, on our website or in public filings. The full text of our code of business conduct and ethics is posted on the investor relations section of our website at https://ir.sutrobio.com/corporate-governance/governance-documents.
Corporate Social Responsibility
We believe that corporate social responsibility initiatives are important to our business and to creating sustainable value for our stockholders and wider stakeholder group. Our Board of Directors and management are committed to these initiatives and believe these efforts will benefit our employees, partners, and the communities in which we operate.
Insider Trading Policy and Anti-hedging
Compensation Committee Interlocks and Insider Participation
During 2025, Mr. Lobacki, Mr. Petree, and Dr. Wigginton served on our Compensation Committee. None of our current executive officers has served as a member of the Board of Directors, or as a member of the Compensation or similar Committee, of any entity that has one or more executive officers who served on our Board of Directors or Compensation Committee during the fiscal year ended December 31, 2025.
Board and Committee Meetings and Attendance
The Board of Directors and its Committees meet regularly throughout the year and also hold special meetings and act by written consent from time to time. During 2025, the Board of Directors held twelve meetings including video conference meetings; the Audit Committee held four meetings; the Compensation Committee held nine meetings; the Science and Technology Committee held four meetings; and the Nominating and Governance Committee held five meetings. During 2025, none of the directors attended fewer than 75% of the aggregate of the total number of meetings held by the Board of Directors during his or her tenure and the total number of meetings held by all Committees of the Board of Directors on which such director served during his or her tenure. The independent members of the Board of Directors also meet separately without management on a regular basis to discuss such matters as the independent directors consider appropriate.
Board Attendance at Annual Stockholders’ Meeting
We invite and encourage each member of our Board of Directors to attend our annual meetings of stockholders. All of our directors attended our 2025 annual meeting of shareholders, which was held virtually. We do not have a
9
formal policy requiring attendance of our annual meetings of stockholders by the members of our Board of Directors.
Communication with Directors
Stockholders and interested parties who wish to communicate with our Board of Directors, non-management members of our Board of Directors as a group, a Committee of the Board of Directors or a specific member of our Board of Directors (including our Chair) may do so by letters addressed to:
Sutro Biopharma, Inc.
c/o Corporate Secretary
111 Oyster Point Boulevard
South San Francisco, California, 94080
All communications by letter addressed to the attention of our Corporate Secretary will be reviewed by the Corporate Secretary and provided to the members of the Board of Directors unless such communications are unsolicited items, sales materials and other routine items and items unrelated to the duties and responsibilities of the Board of Directors.
Considerations in Evaluating Director Nominees
The Nominating and Governance Committee is responsible for identifying, considering and recommending candidates to the Board of Directors for Board membership. A variety of methods are used to identify and evaluate director nominees, with the goal of maintaining and further developing a diverse, experienced and highly qualified Board of Directors. Candidates may come to our attention through current members of our Board of Directors, professional search firms, stockholders or other persons.
The Nominating and Governance Committee will recommend to the Board of Directors for selection all nominees to be proposed by the Board of Directors for election by the stockholders, including approval or recommendation of a slate of director nominees to be proposed by the Board of Directors for election at each annual meeting of shareholders, and will recommend all director nominees to be appointed by the Board of Directors to fill interim director vacancies.
Our Board of Directors encourages selection of directors who will contribute to the company’s overall corporate goals. The Nominating and Governance Committee may from time-to-time review and recommend to the Board of Directors the desired qualifications, expertise and characteristics of director candidates, including such factors as business experience, diversity of viewpoints and experience, and personal skills in life sciences and biotechnology, finance, marketing, financial reporting and other areas that are expected to contribute to an effective Board of Directors. Exceptional candidates who do not meet all of these criteria may still be considered. In evaluating potential candidates for the Board of Directors, the Nominating and Governance Committee considers these factors in light of the specific needs of the Board of Directors at that time.
In addition, under our Corporate Governance Guidelines, a director is expected to spend the time and effort necessary to properly discharge such director’s responsibilities. Accordingly, a director is expected to regularly attend meetings of the Board of Directors and Committees on which such director sits, and to review prior to meetings material distributed in advance for such meetings. Thus, the number of other public company boards and other boards (or comparable governing bodies) on which a prospective nominee is a member, as well as his or her other professional responsibilities, will be considered. Also, under our Corporate Governance Guidelines, there are no limits on the number of three-year terms that may be served by a director. However, in connection with evaluating recommendations for nomination for reelection, the Nominating and Governance Committee considers director tenure. We value diverse viewpoints and experiences on a company-wide basis but have not adopted a specific policy regarding Board diversity.
10
Stockholder Recommendations for Nominations to the Board of Directors
The Nominating and Governance Committee will consider properly submitted stockholder recommendations for candidates for our Board of Directors who meet the minimum qualifications as described above. The Nominating and Governance Committee does not intend to alter the manner in which it evaluates candidates, including the minimum criteria set forth above, based on whether or not the candidate was recommended by a stockholder. A stockholder of record can nominate a candidate for election to the Board of Directors by complying with the procedures in Article I, Section 1.12 of our Amended and Restated Bylaws. Any eligible stockholder who wishes to submit a nomination should review the requirements in the Amended and Restated Bylaws on nominations by stockholders.
Any nomination should be sent in writing to our Corporate Secretary, Sutro Biopharma, Inc., 111 Oyster Point Boulevard, South San Francisco, California, 94080. Submissions must include the full name of the proposed nominee, complete biographical information, a description of the proposed nominee’s qualifications as a director, other information regarding the nominee and proposing stockholder as specified in our Amended and Restated Bylaws, and certain representations regarding the nomination. Any such submission must be accompanied by the written consent of the proposed nominee to be named as a nominee and to serve as a director if elected. These candidates are evaluated at meetings of the Nominating and Governance Committee and may be considered at any point during the year. If any materials are provided by a stockholder in connection with the recommendation of a director candidate, such materials are forwarded to the Nominating and Governance Committee.
Additional information regarding the process for properly submitting stockholder nominations for candidates for membership on our Board of Directors is set forth below under “Stockholder Proposals to Be Presented at Next Annual Meeting.” In addition to satisfying the foregoing requirements under our Amended and Restated Bylaws, to comply with the universal proxy rules, stockholders who intend to solicit proxies in support of director nominees other than the Company’s nominees for the 2027 Annual Meeting of Shareholders must provide notice that sets forth the information required by Rule 14a-19 under the Exchange Act.
11
PROPOSAL NO. 1
ELECTION OF CLASS II DIRECTORS
Our Board of Directors is divided into three classes. Each class serves for three years, with the terms of office of the respective classes expiring in successive years. Directors and director nominees in Class II will stand for election at this meeting. The terms of office of directors in Class III and Class I do not expire until the annual meetings of stockholders to be held in 2027 and 2028, respectively. Our Nominating and Governance Committee recommended to our Board of Directors, and our Board of Directors nominated Ms. Chung, Ms. Matsui, and Mr. Panek, each an incumbent Class II director, for election as Class II directors at the Annual Meeting. At the recommendation of our Nominating and Governance Committee, our Board of Directors proposes that each of the Class II nominees be elected as a Class II director for a three-year term expiring at the annual meeting of shareholders to be held in 2029 and until such director’s successor is duly elected and qualified or until such director’s earlier resignation or removal.
Each director will be elected by a plurality of the votes cast by the holders of the shares present in person or represented by proxy at the meeting and entitled to vote on the election of directors (Broker non‑votes and “WITHHOLD” votes have no effect on the outcome.). This means that the three individuals nominated for election to the Board of Directors at the Annual Meeting receiving the highest number of “FOR” votes will be elected. You may either vote “FOR” one, two, or all of the nominees or “WITHHOLD” your vote with respect to one, two, or all of the nominees. A “WITHHOLD” vote will have no effect on the outcome. Shares represented by proxies will be voted “FOR” the election of each of the Class II nominees, unless the proxy is marked to withhold authority to so vote. You may not cumulate votes in the election of directors. If any nominee for any reason is unable to serve, the proxies may be voted for such substitute nominee as the proxy holders, who are officers of our company, might determine. Each nominee has consented to being named in this proxy statement and to serve if elected. Proxies may not be voted for more than three directors.
Nominees to the Board of Directors
The nominees and their ages as of March 31, 2026 are provided in the table below. Additional biographical information for each nominee is set forth in the text below the table.
Name |
|
Age |
|
Class |
Jane Chung, R.Ph. |
|
55 |
|
Class II Director |
Connie Matsui(1) |
|
72 |
|
Class II Director |
James Panek(1)(2) |
|
73 |
|
Class II Director |
(1) Member of our Nominating and Governance Committee
(2) Member of our Science and Technology Committee
Jane Chung, R.Ph., has served as our Chief Executive Officer and a member of our Board of Directors since March 2025. From May 2015 to August 2021, Ms. Chung served in several leadership roles at AstraZeneca, including as President and General Manager of AstraZeneca Canada, Vice President of Sales and Marketing of U.S. Immuno-Oncology, and Senior Commercial Business Director. Prior to that, from May 2013 to May 2015, Ms. Chung served as a Regional Sales Director and Director of Sales Productivity and Effectiveness for Onyx Pharmaceuticals Inc. From October 2003 to May 2013, she served in various commercial roles for Genentech, Inc., including as Commercial Operations Manager, Division Manager and Senior Marketing Manager. Ms. Chung served on the Board of Directors of Viracta Therapeutics, Inc. from 2022 to 2024 and serves on non-profit boards in the science, education, and community development arenas. Ms. Chung received a B.S. in Pharmacy from St. John's University and a B.A. in Psychology from Columbia University. We believe that Ms. Chung is qualified to serve on our Board
12
of Directors because of her role as our Chief Executive Officer as well as her prior experience at various roles within Sutro Biopharma and her broad experience in the biotechnology industry.
Connie Matsui has served as a member, and Chair, of our Board of Directors since June 2019 and brings over 18 years of general management experience in the biotechnology industry. From 2004 to 2009, Ms. Matsui served in various leadership positions at Biogen Idec, Inc., including as Executive Vice President, Knowledge and Innovation Networks and Executive Committee member. Prior to that, Ms. Matsui served in various leadership positions at IDEC Pharmaceuticals, a predecessor of Biogen Idec, including Senior Vice President; Collaboration Chair for the late-stage development and commercialization of rituximab (tradenames: Rituxan® and MabThera®) in partnership with Roche and Genentech; and Project Leader for Zevalin®, the first radioimmunotherapy approved by the U.S. FDA. Prior to entering the biotechnology industry, Ms. Matsui worked for Wells Fargo Bank in general management, marketing and human resources. Ms. Matsui currently serves on the board of directors of Artelo Biosciences, Inc., and has served on not-for-profit boards at the local, national and global level. Ms. Matsui previously served on the board of directors of Halozyme Therapeutics, Inc. from July 2006 to February 2025. Ms. Matsui received a B.A. and an M.B.A. from Stanford University. We believe that Ms. Matsui is qualified to serve on our Board of Directors because of her wealth of operational and managerial experience, including her extensive knowledge of the biotechnology industry, her service in other public company management teams and boards and her expertise in organizational and operational development. Her operational development experience includes serving as a Collaboration Chair for the late-stage development and commercialization of rituximab in the three-way IDEC Pharmaceuticals, Roche and Genentech collaboration. Her knowledge in these and other areas provide critical insights to our business, particularly as our Board of Directors considers our collaboration strategies through our proprietary XpressCF® and XpressCF+® platforms.
James Panek has served as a member of our Board of Directors since January 2020. Since 2011, Mr. Panek has served as an Independent Consultant for various biopharmaceutical companies, and currently serves as Acting COO and Director for CHO Plus, a privately owned biotechnology company. From 2010 to 2011, Mr. Panek served as interim President, Chief Executive Officer and Principal Financial Officer at DiaDexus, Inc. From 2007 to 2010, Mr. Panek served as President, Chief Executive Officer and Principal Financial Officer for VaxGen, Inc., now a subsidiary of DiaDexus, Inc. From 2002 to 2006, Mr. Panek served as Senior and Executive Vice President of VaxGen, Inc., and Co-Chief Executive Officer and Chairman of the Board for Celltrion Inc., then a VaxGen manufacturing joint venture in Inchon, Korea. In his role with Celltrion, Mr. Panek was responsible for the development and FDA licensure of the first large scale biopharmaceutical manufacturing facility in Asia. From 1982 to 2001, Mr. Panek served in various capacities with Genentech, Inc., including Senior Vice President, Product Operations, and Vice President, Manufacturing, Engineering and Facilities, where he led the development of the world’s largest biotechnology manufacturing facility and was responsible for all operations involved in supplying products for preclinical, clinical, and commercial use. Prior to joining Genentech, Mr. Panek spent six years with Eli Lilly in a variety of engineering and development positions. Mr. Panek previously served on the boards of directors of DiaDexus, Inc., VaxGen, Inc. and Celltrion Inc. Mr. Panek received a B.S. and an M.S. in Chemical Engineering from the University of Michigan. We believe that Mr. Panek is qualified to serve on our Board of Directors because of his 30 years of leadership experience in the biopharmaceutical industry. In particular, Mr. Panek has broad and deep extensive manufacturing and process development experience in our industry, including leading the construction, qualification and operation of Genentech’s pioneering large scale commercial manufacturing facility and his deep experience in managing supply chains and ensuring supply of preclinical, clinical and commercial products.
Continuing Directors
The directors who are serving for terms that end following the Annual Meeting and their ages as of March 31, 2026 are provided in the table below. Additional biographical information for each nominee is set forth in the text below the table.
13
Name |
|
Age |
|
Class |
Heidi Hunter(1)(2) |
|
67 |
|
Class I Director |
Jon Wigginton, M.D.(1)(3) |
|
64 |
|
Class I Director |
Michael Dybbs, Ph.D.(1) |
|
51 |
|
Class I Director |
Joseph M. Lobacki (2)(3) |
|
67 |
|
Class III Director |
Daniel H. Petree (3)(4) |
|
70 |
|
Class III Director |
Sukhi Jagpal(2) |
|
52 |
|
Class III Director |
(1) Member of our Science and Technology Committee
(2) Member of our Audit Committee
(3) Member of our Compensation Committee
(4) Member of our Nominating and Governance Committee
Heidi Hunter has served as a member of our Board of Directors since November 2021. Ms. Hunter is the former President of Cardinal Health Specialty Solutions, a specialty healthcare business. Prior to Cardinal Health, Ms. Hunter served as Senior Vice President for UCB (Union Chimique Belge), a multinational biopharmaceutical company with a primary focus on neurology and immunology disorders from September 2015 to September 2020. Ms. Hunter also served as Senior Vice President and General Manager of Boehringer Ingelheim, a pharmaceutical company, from 2011 to 2015. Prior to Boehringer Ingelheim, Ms. Hunter held similar roles in sales and marketing at Ciba-Geigy (today part of Novartis) and Wyeth Pharmaceuticals LLC (today part of Pfizer) where she led their oncology business. Ms. Hunter also serves on the Board of Directors of Vicore Pharma Holding AB, Bavarian Nordic and IO Biotech. Ms. Hunter received a B.A in Economics from University of Michigan and a M.B.A. from University of Chicago - Booth School of Business. We believe that Ms. Hunter is qualified to serve on our Board of Directors because of her extensive experience in building and developing talent and high-performance teams in the pharmaceutical sector with the skills, motivation, and culture to achieve sustainable results. Ms. Hunter’s experience in various senior management positions, including as the former President of Cardinal Health Specialty Solutions, a portfolio of specialty healthcare businesses, strengthens our Board of Directors’ ability to oversee the important human capital management element of our company.
Jon Wigginton, M.D., has served as a member of our Board of Directors since November 2020. Dr. Wigginton is a physician and scientist with expertise in cancer immunotherapy drug development. He has over 25 years of experience in clinical oncology, with leadership experience in both academia and industry. Dr. Wigginton currently serves as President, Research and Development at Bright Peak Therapeutics. Most recently, he was Senior Advisor and Chairman of the SAB at Cullinan Oncology, Inc., having served previously as the Chief Medical Officer focused on developing oncology therapeutics, and as an advisor for MPM Capital, a healthcare investment firm from 2020 to 2021. From August 2013 to March 2020, Dr. Wigginton served as Senior Vice President, Clinical Development and Chief Medical Officer for MacroGenics, Inc., a clinical-stage biopharmaceutical company focused on discovering and developing innovative monoclonal antibody-based therapeutics. From 2008 to 2013, Dr. Wigginton served as the Therapeutic Area Head, Immuno-Oncology, Early Clinical Research and Executive Director, Discovery Medicine-Clinical Oncology at Bristol-Myers. From 2006 to 2008, Dr. Wigginton served as the Director of Clinical Oncology at Merck Research Laboratories. During his academic career, Dr. Wigginton has held several positions at the National Cancer Institute Center for Cancer Research, including Head of Investigational Biologics Section, Pediatric Oncology Branch. Dr. Wigginton also served as a member of the Board of Directors of Checkmate Pharmaceuticals from January 2022 through May 2022, when the company was acquired by Regeneron. Dr. Wigginton previously served as President and as a member of the Board of directors of the Society for Immunotherapy of Cancer (non-profit). Dr. Wigginton received an M.D. and B.S. in Biology from the University of Michigan. We believe that Dr. Wigginton is qualified to serve on our Board of Directors because of his years of experience in clinical oncology and immunotherapy drug development. Dr. Wigginton’s significant experience in developing strategies that ultimately resulted in FDA approval of new treatment options offers a valuable perspective to our Board of Directors’ discussions regarding clinical development of new oncology therapeutics.
Michael Dybbs, Ph.D., has served as a member of our Board of Directors since July 2018. Dr. Dybbs is currently a partner at Samsara BioCapital, where he has worked since March 2017. Prior to joining Samsara, Dr. Dybbs was a
14
partner at New Leaf Venture Partners, where he worked from May 2009 until September 2016. Before joining New Leaf Venture Partners, L.L.C., Dr. Dybbs was a principal at the Boston Consulting Group. Dr. Dybbs currently serves on the boards of directors of Nkarta Therapeutics (NKTX), Kalaris Therapeutics (KLRS), Spyglass Pharma (SGP), and several private companies. Dr. Dybbs previously served on the boards of directors of Versartis, Inc. and Dimension Therapeutics, Inc. Dr. Dybbs received an A.B. in biochemical sciences from Harvard College and a Ph.D. in molecular biology from University of California, Berkeley, where he was awarded a Howard Hughes Medical Institute fellowship. We believe that Dr. Dybbs is qualified to serve on our Board of Directors because of his financial and strategic expertise as well as his deep understanding of the life sciences sector developed over years of investing and advising companies of all sizes. Our Board of Directors particularly values Dr. Dybbs’s experience in evaluation of and development of messaging strategies for clinical data.
Joseph M. Lobacki has served as a member of our Board of Directors since February 2017. From January 2020 to January 2024, Mr. Lobacki served as president and CEO of Artax, a private biopharmaceutical company developing treatments for autoimmune and inflammatory diseases focused on modulating the T-Cell Receptor response to antigen stimulation. Since January 2024, Mr. Lobacki has served in an advisory role at Artax, and as an independent consultant to Biotech companies. Previously, Mr. Lobacki served as Executive Vice President and Chief Commercial Officer for Verastem, Inc., a biopharmaceutical company focused on the development and commercialization of therapies for the treatment of hematologic malignancies. From November 2016 to December 2017, Mr. Lobacki served as Chief Operating Officer for Crestovo, a clinical-stage biopharmaceutical company focused on microbiome therapies. From 2014 to 2016, Mr. Lobacki served as Chief Commercial Officer at Medivation, Inc., a biopharmaceutical company focused on development of novel therapies for the treatment of serious diseases. From 2012 to 2014, Mr. Lobacki also served as General Manager of Oncology and an independent biotechnology consultant at Idera Pharmaceuticals, Inc., a biopharmaceutical company focused on therapies for cancer and rare diseases. Previously, Mr. Lobacki served as Senior Vice-President and Chief Commercial Officer at Micromet, Inc., Senior Vice-President and General Manager of US Transplant and Oncology at Genzyme Corporation and in various other positions at SangStat Medical Corporation, Cell Pathways, Inc., Rhone-Poulenc Rorer and Lederle Laboratories. Mr. Lobacki previously served on the Board of Directors of Celator Pharmaceuticals Inc. Mr. Lobacki received a B.S. in Biology from Boston College and a B.S. in Pharmacy from the Massachusetts College of Pharmacy. We believe that Mr. Lobacki is qualified to serve on our Board of Directors because of his extensive biopharmaceutical managerial and commercial experience, including his positions of Chief Commercial Officer, Chief Operating Officer, Executive Vice President and Chief Executive Officer of biopharma companies. His broad understanding of medical affairs and commercial development due to his experiences contributes an important perspective to our Board of Directors’ discussions regarding commercial strategy, business development and human capital management matters.
Daniel H. Petree, has served as a member of our Board of Directors since August 2009. In April 2012, Mr. Petree co-founded Four Oaks Partners Consulting, LLC, which provided transaction advisory services to small and medium-sized life science companies until 2021 and in 2000, Mr. Petree co-founded P2 Partners, LLC, Four Oaks' predecessor in the same business which operated until 2012. Before co-founding P2 Partners, Mr. Petree served as President and Chief Operating Officer of Axys Pharmaceuticals, Inc., Executive Vice President and Chief Financial Officer of Arris Pharmaceuticals, Incorporated and Vice President of Business Development at TSI Corporation and was a corporate and securities lawyer. Mr. Petree previously served on the boards of directors of Lpath, Inc., Biocept, Inc. and Cypress Bioscience, Inc. along with a number of privately held biotechnology companies. Mr. Petree received an A.B. in History and Political Science from Stanford University and a J.D. from the University of Michigan Law School. We believe that Mr. Petree is qualified to serve on our Board of Directors because of his deep knowledge and expertise in structuring and negotiating pharmaceutical partnering arrangements and strategic transactions. His significant business development and financing expertise, and in particular his experience with asset monetization, provides our Board of Directors with valuable insight into financing and growth strategies for us.
Sukhi Jagpal has served as a member of our Board of Directors since August 2024. Mr. Jagpal brings over twenty years of experience across the biotechnology industry. He previously served as the Chief Financial Officer of Sierra Oncology, guiding the company from its initial public offering in 2015 through to the completion of its $1.9 billion acquisition by GSK in 2022. Previously, Mr. Jagpal served as the Chief Financial Officer of QLT Inc., a public biotechnology company focused primarily on ophthalmology in addition to oncology and dermatology, from 2012 to 2015, where he also served as the Senior Director, Finance and Corporate Controller. Prior to joining QLT in 2003, Mr. Jagpal held senior finance positions with Pivotal Corporation, 360networks Inc, and KPMG LLP. Mr. Jagpal is
15
a Chartered Professional Accountant, a Chartered Business Valuator, and holds an M.B.A. from the S.C. Johnson Graduate School of Management, Cornell University. We believe that Mr. Jagpal is qualified to serve on our Board of Directors because his financial and accounting experience in guiding biotechnology companies, and in particular his prior leadership positions in the biotechnology industry, provides valuable insight.
Family Relationships
There are no familial relationships among any of our directors and executive officers.
Non-Employee Director Compensation
Our compensation arrangements for non-employee directors are reviewed periodically by our Compensation Committee and our Board of Directors. In addition, at the Compensation Committee’s direction, FW Cook, the Compensation Committee’s independent compensation consultant, provided a competitive analysis of director compensation levels, practices and design features as compared to the general market as well as to our compensation peer group. The adjustments made in 2025 are noted below.
Our non-employee directors receive the following compensation pursuant to a program adopted by our Board of Directors:
Non-employee directors are also reimbursed for reasonable expenses incurred in serving as a director, including travel expenses for attending meetings of our Board of Directors.
The following table sets forth the compensation earned by or paid to the non-employee directors who served during 2025 for services provided during the year ended December 31, 2025. Mr. Newell, our former Chief Executive Officer, and Ms. Chung, our current Chief Executive Officer, each received no compensation for his or her service as a director during 2025.
16
Name |
|
Fees Earned or |
|
Option Awards ($)(1) |
|
Total ($) |
|
||||||
|
|
Paid in Cash ($) |
|
|
|
|
|
||||||
Connie Matsui |
|
$ |
|
80,000 |
|
$ |
|
41,795 |
|
$ |
|
121,795 |
|
Michael Dybbs, Ph.D. |
|
|
|
45,000 |
|
|
|
41,795 |
|
|
|
86,795 |
|
John Freund, M.D.(2) |
|
|
|
21,703 |
|
|
|
— |
|
|
|
21,703 |
|
Heidi Hunter |
|
|
|
57,500 |
|
|
|
41,795 |
|
|
|
99,295 |
|
Joseph Lobacki |
|
|
|
67,000 |
|
|
|
41,795 |
|
|
|
108,795 |
|
James Panek |
|
|
|
55,000 |
|
|
|
41,795 |
|
|
|
96,795 |
|
Daniel Petree |
|
|
|
53,500 |
|
|
|
41,795 |
|
|
|
95,295 |
|
Jon Wigginton, M.D. |
|
|
|
58,500 |
|
|
|
41,795 |
|
|
|
100,295 |
|
Sukhi Jagpal |
|
|
|
57,500 |
|
|
|
41,795 |
|
|
|
99,295 |
|
(1) The amounts reported in this column represent the aggregate grant date fair value of the stock options granted to our directors during the year ended December 31, 2025 as computed in accordance with Financial Accounting Standards Board Accounting Standards Codification (FASB ASC) Topic 718. The assumptions used in calculating the aggregate grant date fair value of the stock options reported in this column are set forth in Note 13 to our financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2025. The amounts reported in this column reflect the accounting cost for these stock options, and do not correspond to the actual economic value that may be received by our directors from the stock options. For information regarding the number of stock options held by each non-employee director as of December 31, 2025, see the table below.
(2) Dr. Freund resigned from our Board of Directors in June 2025.
The following table sets forth the number of outstanding equity awards held by the non-employee directors who served during 2025 as of December 31, 2025:
Name |
|
Option Awards |
|
Connie Matsui |
|
|
19,706 |
Michael Dybbs, Ph.D. |
|
|
21,163 |
John Freund M.D. |
|
|
15,663 |
Heidi Hunter |
|
|
17,738 |
Joseph Lobacki |
|
|
22,797 |
James Panek |
|
|
19,799 |
Daniel Petree |
|
|
21,163 |
Jon Wigginton M.D. |
|
|
18,599 |
Sukhi Jagpal |
|
|
12,895 |
OUR BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THE ELECTION OF EACH OF THE NOMINATED CLASS II DIRECTORS.
17
PROPOSAL NO. 2
RATIFICATION OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Our Audit Committee has selected Ernst & Young LLP as our principal independent registered public accounting firm to perform the audit of our financial statements for the fiscal year ending December 31, 2026. Ernst & Young LLP audited our financial statements for the fiscal years ended December 31, 2025 and 2024. We expect that representatives of Ernst & Young LLP will be present at the Annual Meeting, will be able to make a statement if they so desire and will be available to respond to appropriate questions.
At the Annual Meeting, the stockholders are being asked to ratify the appointment of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026. Although ratification by stockholders is not required by law, our Audit Committee is submitting the selection of Ernst & Young LLP to our stockholders because we value our stockholders’ views on our independent registered public accounting firm and as a matter of good corporate governance. If this proposal does not receive the affirmative approval of a majority of the votes cast on the proposal, the Audit Committee would reconsider the appointment. Notwithstanding its selection and even if our stockholders ratify the selection, our Audit Committee, in its discretion, may appoint another independent registered public accounting firm at any time during the year if the Audit Committee believes that such a change would be in our best interests and the interests of our stockholders.
The following table presents fees for professional audit services rendered by Ernst & Young LLP for the audit of our annual financial statements for the years ended December 31, 2025 and 2024.
Principal Accountant Fees and Services
Fees Billed |
|
Fiscal Year 2025 |
|
|
Fiscal Year 2024 |
|
||||
Audit fees(1) |
|
$ |
|
1,255,248 |
|
|
$ |
|
2,327,976 |
|
Audit-related fees |
|
|
— |
|
|
|
— |
|
||
Tax fees(2) |
|
|
|
15,750 |
|
|
|
|
189,833 |
|
All other fees |
|
— |
|
|
— |
|
||||
Total fees |
|
$ |
|
1,270,998 |
|
|
$ |
|
2,517,809 |
|
(1) “Audit fees” include fees for professional services provided by Ernst & Young LLP in connection with the audit of our financial statements, review of our quarterly financial statements, and related services that are typically provided in connection with registration statements, including the registration statement for our public offerings.
(2) “Tax fees” include fees for tax compliance and advice. Tax advice fees encompass a variety of permissible services, including technical tax advice related to federal and state income tax matters, assistance with sales tax, and assistance with tax audits.
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Registered Public Accounting Firm
Our Audit Committee generally pre-approves all audit and permissible non-audit services provided by the independent registered public accounting firm. These services may include audit services, audit-related services, tax services and other services. Pre-approval is detailed as to the particular service or category of services and is generally subject to a specific budget. The independent registered public accounting firm and management are required to periodically report to the Audit Committee regarding the extent of services provided by the independent registered public accounting firm in accordance with this pre-approval, and the fees for the services performed to date. Our Audit Committee may also pre-approve particular services on a case-by-case basis. All of the services relating to the fees described in the table above were approved by our Audit Committee.
OUR BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” APPROVAL OF PROPOSAL NO. 2.
18
REPORT OF THE AUDIT COMMITTEE
The information contained in the following report of the Audit Committee is not considered to be “soliciting material,” “filed” or incorporated by reference in any past or future filing by us under the Securities Exchange Act of 1934, as amended, or the Securities Act of 1933, as amended, unless and only to the extent that we specifically incorporate it by reference.
The Audit Committee has reviewed and discussed with our management and Ernst & Young LLP our audited financial statements as of and for the year ended December 31, 2025. The Audit Committee has also discussed with Ernst & Young LLP the matters required to be discussed by the applicable requirements of the Public Company Accounting Oversight Board (United States) and the U.S. Securities and Exchange Commission.
The Audit Committee has received and reviewed the written disclosures and the letter from Ernst & Young LLP required by applicable requirements of the Public Company Accounting Oversight Board regarding the independent accountant’s communications with the Audit Committee concerning independence and has discussed with Ernst & Young LLP its independence.
Based on the review and discussions referred to above, the Audit Committee recommended to our Board of Directors that the audited financial statements as of and for the year ended December 31, 2025 be included in our Annual Report on Form 10-K for the year ended December 31, 2025 for filing with the U.S. Securities and Exchange Commission.
Submitted by the Audit Committee
Sukhi Jagpal, Chair
Heidi Hunter
Joseph Lobacki
19
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information with respect to the beneficial ownership of our common stock as of March 31, 2026, by:
Percentage ownership of our common stock is based on 16,567,762 shares of our common stock outstanding on March 31, 2026. We have determined beneficial ownership in accordance with the rules of the SEC, and thus it represents sole or shared voting or investment power with respect to our securities, and the information is not necessarily indicative of beneficial ownership for any other purpose. Unless otherwise indicated below, to our knowledge, the persons and entities named in the table have sole voting and sole investment power with respect to all shares that they beneficially owned, subject to community property laws where applicable. We have deemed all shares of common stock subject to options or other convertible securities held by that person or entity that are currently exercisable or that will become exercisable within 60 days of March 31, 2026 to be outstanding and to be beneficially owned by the person or entity holding the option for the purpose of computing the percentage ownership of that person or entity but have not treated them as outstanding for the purpose of computing the percentage ownership of any other person or entity. Unless otherwise indicated, the address of each beneficial owner listed in the table below is c/o Sutro Biopharma, Inc., 111 Oyster Point Boulevard, South San Francisco, California 94080.
|
|
Beneficial Ownership |
|
|||||
Name of Beneficial Owner |
|
Number |
|
|
Percent |
|
||
5% Stockholders |
|
|
|
|
|
|
||
Affiliates of Biotechnology Value Fund LP(1) |
|
|
1,251,788 |
|
|
|
7.6 |
% |
Directors and Named Executive Officers: |
|
|
|
|
|
|
||
Connie Matsui(2) |
|
|
24,039 |
|
|
* |
|
|
Michael Dybbs, Ph.D.(3) |
|
|
20,496 |
|
|
* |
|
|
Heidi Hunter(4) |
|
|
17,071 |
|
|
* |
|
|
Joseph Lobacki(5) |
|
|
22,130 |
|
|
* |
|
|
James Panek(6) |
|
|
19,132 |
|
|
* |
|
|
Daniel Petree(7) |
|
|
22,741 |
|
|
* |
|
|
Jon M. Wigginton, M.D.(8) |
|
|
17,932 |
|
|
* |
|
|
Sukhi Jagpal(9) |
|
|
10,769 |
|
|
* |
|
|
William J. Newell(10) |
|
|
128,194 |
|
|
* |
|
|
Jane Chung(11) |
|
|
75,080 |
|
|
* |
|
|
Hans-Peter Gerber, Ph.D.(12) |
|
|
30,747 |
|
|
* |
|
|
Gregory Chow(13) |
|
|
4,745 |
|
|
* |
|
|
All executive officers and directors as a group (12 persons) (14) |
|
|
295,143 |
|
|
|
1.8 |
% |
* Represents beneficial ownership of less than one percent.
ownership as of February 10, 2026. According to the statement, (i) BVF directly beneficially owned 668,693 shares of the Issuer's Common Stock, (ii) BVF2 directly beneficially owned 488,916 shares and (iii) Trading Fund OS beneficially owned 75,359 shares. BVF GP, as the general partner of BVF, may be deemed to indirectly beneficially own the 668,693 shares held by BVF. BVF2 GP, as the general partner of BVF2, may be deemed to indirectly beneficially own the 488,916 shares held by BVF2. Partners OS, as the general partner of Trading Fund OS, may be deemed to indirectly beneficially own the 75,359 shares beneficially owned by Trading Fund OS. BVF GPH, as the sole member of each of BVF GP and
20
21
EXECUTIVE OFFICERS
The following table provides information regarding our executive officers as of March 31, 2026:
Name |
|
Age |
|
Position(s) |
Jane Chung, R.Ph. |
|
55 |
|
Chief Executive Officer and Director |
Gregory Chow |
|
53 |
Chief Financial Officer |
|
Hans-Peter Gerber, Ph.D. |
|
63 |
|
Chief Scientific Officer |
David Pauling |
52 |
|
Chief Administrative Officer and General Counsel |
|
Jane Chung, R.Ph. has served as our Chief Executive Officer and a member of our Board of Directors since March 2025. Ms. Chung’s biographical information is set forth above under the heading “Proposal No. 1 Election of Class II Directors - Nominees to the Board of Directors.”
Hans-Peter Gerber, Ph.D. has served as our Chief Scientific Officer since September 2023. From April 2022 to August 2023, Dr. Gerber served as Chief Scientific Officer and Co-founder of Codeable Therapeutics, an ADC startup company. From April 2018 to March 2022, Dr. Gerber served as Chief Scientific Officer and Senior Vice President of 3T Biosciences, Inc. From November 2016 to March 2018, Dr. Gerber served as President and Chief Scientific Officer of Maverick Therapeutics, Inc., which was acquired by Takeda Pharmaceutical Company in 2021. Prior to his service at Maverick, Dr. Gerber held leadership positions at several top biopharmaceutical companies, including Genentech, Seattle Genetics, and Pfizer, where he led the R&D group as Chief Scientific Officer. Dr. Gerber earned his M.S. in Biochemistry and his Ph.D in Molecular Biology from the University of Zurich, Switzerland.
Gregory Chow has served as our Chief Financial Officer since June 2025. Prior to joining Sutro, Mr. Chow served as Chief Financial and Business Officer of NodThera, a biotechnology company, from August 2024 to March, 2025. From January 2024 to August 2024, Mr. Chow served as Chief Financial Officer for Freenome Holdings, Inc., a biotechnology company. From March 2021 to December 2024, Mr. Chow served as Chief Financial Officer of Frontier Medicines Corp., a biopharmaceutical company. Prior to that, Mr. Chow was Executive Vice President and Chief Financial Officer of Aptose Biosciences Inc., a clinical-stage biopharmaceutical company, from November 2013 to March 2021. Earlier in his career, Greg spent fourteen years in investment banking, including leadership roles as Managing Director and Director of Private Placements at Wedbush Securities, and senior roles at RBC Capital Markets and Wells Fargo Securities. Mr. Chow is a Certified Public Accountant (inactive) in the State of California. Mr. Chow received his MBA in Finance from The Wharton School at the University of Pennsylvania, and his BA in Business Economics with an emphasis in Accounting from the University of California, Santa Barbara.
David Pauling, J.D., M.A., has served as our Chief Administrative Officer and General Counsel since April 2025. From February 2021 until March 2025, Mr. Pauling served as our General Counsel. Prior to that, from February 2018 to February 2021, he served as our Executive Director of Legal Affairs. From October 2014 to February 2018, Mr. Pauling served as our Associate General Counsel and Senior IP Counsel and from December 2011 to October 2014, as our IP Counsel. Prior to joining the Company, Mr. Pauling served as an associate attorney at Jones Day from January 2004 to December 2011 and as an associate attorney at Pennie & Edmonds, LLP from October 2002 to December 2003. Mr. Pauling currently serves on the board of directors of CuriOdyssey, a non-profit organization.
22
Mr. Pauling received his J.D. from Santa Clara University School of Law, and an M.A. in Molecular Biology and a B.S. in Biochemistry and Molecular Biology from the University of California, Santa Cruz.
23
EXECUTIVE COMPENSATION
Overview and Narrative
This overview and narrative (“Compensation Disclosure”) describes the key elements of our executive compensation program and compensation decisions for our named executive officers, or NEOs, for 2025. This Compensation Disclosure is intended to be read in conjunction with the tables that immediately follow this section, which provide additional compensation information for our named executive officers. We are a “smaller reporting company” as defined under Rule 405 of the Securities Act of 1933, as amended, and, as such, we are not required to provide the additional disclosures included in this Compensation Disclosure. However, we believe the additional narrative disclosure with respect to our executive compensation program will provide our stockholders with further information regarding our company and our executive compensation program and practices and therefore will assist in their consideration of Proposal No. 3, the non-binding vote with respect to named executive officer compensation.
Our executive compensation programs are designed to attract, motivate and retain qualified and talented executives, incentivize them to achieve our business objectives, and reward them for superior short- and long-term performance and results. The Compensation Committee of the Board of Directors, with input from its independent compensation consultant, oversees these programs and determines compensation for our NEOs.
Our 2025 NEOs were:
NEO |
|
Class |
William J. Newell(1) |
|
Former Chief Executive Officer |
Jane Chung, R.Ph.(2) |
|
Chief Executive Officer |
Hans-Peter Gerber, Ph.D. |
|
Chief Scientific Officer |
Gregory Chow(3) |
|
Chief Financial Officer |
Executive Summary
We are a clinical stage oncology company developing site-specific and novel-format antibody drug conjugates, or ADCs, enabled by our proprietary integrated cell-free protein synthesis platform, XpressCF®, and our site-specific conjugation platform, XpressCF+®. We aim to design and develop therapeutics using the most relevant and potent modalities, including ADCs, bispecific ADCs, immunostimulatory ADCs, or iADCs, and dual payload ADCs, or dpADCs. Our molecules are directed primarily against clinically validated targets where the current standard of care is suboptimal. We believe that our platform allows us to accelerate the discovery and development of potential first-in-class and/or best-in-class molecules by enabling the rapid and systematic evaluation of protein structure-activity relationships to create optimized homogeneous product candidates. Our mission is to transform the lives of patients by creating medicines with improved therapeutic profiles for areas of unmet need.
Our NEO compensation program in 2025 was designed to be aligned with industry practices and align executive compensation with our performance, with an emphasis on long-term equity compensation in the form of stock options and restricted stock units, or RSUs.
Business Highlights
In 2025, our Board of Directors in consultation with our leadership team decided to deprioritize luvelta development and focus on our preclinical portfolio, including STRO-004, STRO-006, and STRO-227. In doing so, we refocused on developing our pipeline and platform, delivering on key milestones, furthering our capabilities with strong partner relationships, right-sizing the organization, and strengthening our financial health.
24
We have a broad preclinical and clinical pipeline with multiple assets deployed across multiple oncology indications. Based on our proprietary XpressCF® and XpressCF+® platforms, we have also entered into multi-target, product-focused collaborations with leaders in the field of oncology. In 2025, we simultaneously wound down luvelta development, advanced our preclinical programs, including beginning clinical development of our lead wholly-owned product candidate, STRO-004, a single homogeneous ADC directed against tissue factor, or TF, which we are developing for the treatment of solid tumors. We also continued to support the development of partnered product candidates in clinical trials and advanced preclinical assets, including STRO-227 and STRO-006 towards the clinic.
Key 2025 achievements reflected significant scientific, clinical, and business performance that we believe can create long-term value for our stockholders, including the following:
25
Compensation Practices and Governance Highlights |
|
Pay for Performance |
Significant link between the compensation of our NEOs and the achievement of our short- and long-term business objectives through annual cash incentives that are tied to progression of our proprietary and partnered pipeline products, deal-making, financial performance, manufacturing success, and cultural and compliance priorities and with long-term equity compensation that rewards creation of stockholder value through stock options and RSUs |
Stockholder Alignment |
Alignment of the interests of our NEOs with those of our stockholders through the use of long-term equity incentives, the value of which is dependent on our stock price performance |
Compensation Governance |
Independent directors on the Compensation Committee Compensation Committee meets regularly in executive session without management present Independent compensation consultant, FW Cook, reports directly to the Compensation Committee Compensation Committee regularly considers risks related to compensation policies and practices |
Change in Control Provisions |
Change in control payments contingent upon “double-trigger” No tax gross-ups on severance or change in control benefits |
Post-Termination Retirement Benefits |
No post-termination retirement or pension benefits |
Prohibition on Hedging, Margin Loans and Pledging |
Prohibit hedging, purchases on margin, and pledging of our common stock by all employees and directors |
Clawback Policy |
Maintain a compensation recovery policy |
Stockholder Advisory Vote
At our annual meeting of shareholders on June 5, 2026, we will again conduct a non-binding stockholder advisory vote on the compensation of our named executive officers (commonly known as a “Say-on-Pay” vote). We conducted a Say-on-Pay vote in 2025, in which the stockholders approved on an advisory basis the compensation of our named executive officers with a 77.7% support rate. While we viewed this result as supportive of our compensation program, we recognized that approximately 22.3% of votes were not cast in favor, and we took proactive steps to understand our stockholders’ perspectives. Following the 2025 annual meeting, members of our management team reached out to certain stockholders and engaged in direct conversations with these stockholders on a range of topics, including our executive compensation philosophy, the structure and weighting of our annual incentive program, and the relationship between pay and our corporate performance. We value the opinions of our stockholders, and the Compensation Committee and the Board of Directors considered the outcome of the advisory vote and discussions with our stockholders, and will continue to do so in the future, including the vote which will take place at the 2026 annual meeting, when making compensation decisions for our NEOs.
Compensation Philosophy, Objectives and Development
Our 2025 compensation strategy emphasizes pay for performance by using both performance-based annual cash incentives and long-term equity awards in the form of stock options and RSUs. Stock options are utilized because they only provide value if the stock price increases. RSUs are also used to retain talent, align with long-term value
26
(both increases and reductions), and because RSUs are less dilutive than stock options. Our stock options generally vest monthly over four years; with a one-year cliff vesting associated with a new hire grant, and our RSUs generally vest annually over four years (with 25% vesting annually), to align with long-term value creation as well as our clinical development and commercialization timeline.
Consistent with our pay-for-performance philosophy, and the long product development life cycles in the biopharmaceuticals industry, the Compensation Committee links the compensation of our executive officers to performance by emphasizing equity compensation opportunities for long-term performance and cash incentives for near-term goal alignment. Thus, the total compensation provided to our executive officers will vary from year to year and will vary between executive officers based on corporate performance, including performance against annual goals that are pre-established by the Compensation Committee, as well as individual performance. The Compensation Committee’s 2025 compensation decisions reflected, among other things, the level of goal achievement related to the potential for long-term value creation from our platform and clinical, manufacturing and business achievements. The Compensation Committee will continue to consider scientific and business achievements and market capitalization, among the many factors considered, when making compensation decisions.
Our NEOs are also provided market-competitive health and welfare benefits, and as described below, they may be entitled to receive additional benefits if certain criteria are met at termination of employment.
We will continue to refine the design of our compensation program and implement one that is appropriate for our company given our business, industry, growth, and other factors.
Program Development and Role of Compensation Committee, Compensation Consultant and Management
Role of Compensation Committee. Our Compensation Committee is responsible for overseeing the total compensation of our executive officers, including the underlying philosophy and related policies. As such, the Compensation Committee designs, implements, reviews and approves all compensation for our CEO and our other executive officers. From time to time, and in conjunction with our Compensation Committee, the independent members of our Board of Directors, including our Chairperson, may also be involved in setting the compensation of our CEO and other executive officers and determining the corporate objectives upon which our short-term incentives are based and assessed. Our Compensation Committee’s decisions and recommendations regarding executive compensation are based on the Compensation Committee’s assessment of the performance of our company and each individual executive officer, as well as other factors, such as prevailing industry trends, retention, and the competitive market for executive talent.
The Compensation Committee consults with its independent compensation consultant, legal counsel and other advisors in designing our compensation program, including in evaluating the competitiveness of individual compensation and in relation to our performance goals. The Compensation Committee also considers peer company data and factors such as the past, current and expected contributions of each NEO, our corporate performance and strategic focus, global economic conditions, the mix of compensation that would be most appropriate for each NEO, and such officer’s particular responsibilities, experience, level of accountability and decision authority.
Role of CEO and Management. As part of the process for setting the compensation of our NEOs, our Chief Executive Officer provides the Compensation Committee with her performance assessments of the company and of the individual NEOs and recommends to the Compensation Committee base salaries, target annual cash incentives (as a percentage of base salary), annual cash incentive payouts (actual incentive paid based on performance against goals), and stock-based compensation for our NEOs (other than for herself). The Compensation Committee considers our Chief Executive Officer’s input and can accept, reject or modify these recommendations in its discretion.
The Compensation Committee meets in executive session without management. Various members of management may attend Committee meetings, and they and other employees, as well as outside advisors or consultants, may be invited by the Compensation Committee to make presentations, provide financial, competitive market, or other background information or advice. None of our NEOs were present during the Compensation Committee’s determinations regarding his or her own compensation.
27
Role of Independent Compensation Consultant & Market Data. The Compensation Committee has retained FW Cook as its compensation consultant. FW Cook reports directly to the Compensation Committee and takes its direction from the Chair of the Compensation Committee, working with management on select issues under the Compensation Committee’s oversight. The Compensation Committee retained FW Cook in 2025 to provide data, context, and advice regarding executive officer compensation and our peer group, and to assist with compensation risk assessments.
Peer Groups Used in Program Development and Compensation Decisions
Our Compensation Committee considers peer data as one factor in its overall compensation analysis and for purposes of assessing the competitiveness of the executive compensation program. An individual NEO may earn more or less than the peer group median depending on factors described below under the heading “2025 Compensation Decisions,” including the individual’s experience, role, and past and expected future performance.
Fiscal 2025 Peer Group
In the third quarter of 2024, the Compensation Committee reviewed and updated our peer group to include the group of companies set forth below based on, among other considerations, objective size criteria, including industry, market capitalization, headcount, pipeline, location, scope of operations and commercial stage. We refer to this peer group of 18 companies as the “2025 Peer Group.” When this peer group was approved by the Compensation Committee to inform 2025 compensation, these companies had July 15, 2024 market capitalizations of between $54 million and $1.7 billion, and our $345 million market capitalization on this date was within the peer range.
Fiscal 2025 Peer Group |
||||
Alector, Inc. |
|
G1 Therapeutics, Inc. |
|
Mersana Therapeutics, Inc. |
Allogene Therapeutics, Inc. |
|
Gossamer Bio, Inc. |
|
Nurix Therapeutics, Inc. |
Anaptys Bio, Inc. |
|
Gritstone Bio, Inc. |
|
Poseida Therapeutics, Inc. |
Arcus Biosciences, Inc. |
|
IGM Biosciences, Inc. |
|
Replimune Group, Inc. |
Atara Biotherapeutics, Inc. |
|
Kura Oncology, Inc. |
|
Sangamo Therapeutics, Inc. |
Caribou Biosciences, Inc. |
|
MacroGenics, Inc. |
|
Zymeworks Inc. |
Compared to the 2025 Peer Group, the following companies were removed due to acquisition or wind-down of those organizations: Alaunos Therapeutics, Inc., ImmunoGen, Inc., and Jounce Therapeutics, Inc. Two companies that fit the Compensation Committee’s desired criteria and had market capitalization on July 15, 2024 ranging from $192 million to $330 million were added to the 2025 Peer Group: Caribou Biosciences, Inc. and Poseida Therapeutics, Inc.
Compensation Consultant Conflict of Interest Analysis
The Compensation Committee has determined that the work of FW Cook and the individual compensation advisors employed by FW Cook does not create any conflict of interest. In making that determination, the Compensation Committee took into consideration the following factors: (i) the provision of other services to us by FW Cook; (ii) the amount of fees we paid FW Cook as a percentage of FW Cook’s total revenue; (iii) FW Cook’s policies and procedures that are designed to prevent conflicts of interest; (iv) any business or personal relationship of FW Cook or the individual compensation advisors employed by FW Cook with a Sutro executive officer; (v) any business or personal relationship of the individual compensation advisors with any member of the Compensation Committee; and (vi) any Sutro stock owned by FW Cook or the individual compensation advisors employed by the consultant. During 2025, we paid FW Cook fees that constituted less than 1% of FW Cook’s total revenue.
28
2025 Compensation Decisions
Summary
Most 2025 compensation decisions were made in the first quarter of 2025 and were significantly influenced by our scientific and business achievements together with the competitive market for experienced talent. Our 2025 compensation decisions were based on a review of individual performance and potential; achievement of the Company’s corporate goals, including advancing our research and clinical pipelines and financial performance; and a market analysis of competitive compensation based on our 2025 peer group.
Following are highlights of the 2025 compensation decisions:
Base Salary
The purpose of base salary is to provide fixed compensation to attract, retain and motivate executives with the qualifications desired for the individual position. The base salary we provide each of our NEOs is influenced by a number of factors, including the individual’s position, scope of responsibilities, breadth and depth of experience, performance to date, expected future contribution, and their overall mix of base salary, performance-based cash incentives and equity compensation.
The 2025 base salaries for our NEOs were as follows:
NEO |
|
2024 Annual Base Salary |
|
|
2025 Annual Base Salary |
|
|
Increase (%) |
|
|||
Jane Chung, R.Ph. |
|
$ |
560,000 |
|
|
$ |
675,000 |
|
(1) |
|
21 |
% |
Hans-Peter Gerber, Ph.D. |
|
|
515,000 |
|
|
|
530,039 |
|
|
|
3 |
% |
Gregory Chow |
|
|
— |
|
|
|
475,000 |
|
(2) |
N/A |
|
|
William J. Newell |
|
|
711,000 |
|
|
|
711,000 |
|
(3) |
|
0 |
% |
Performance-Based Cash Incentives
During 2025, all of our NEOs were eligible for annual performance-based cash incentive bonuses, which were designed to reward achievement of certain annual corporate goals that the Company believes will ultimately drive stockholder value. Each performance-based cash incentive bonus opportunity is expressed as a target percentage of annual base salary. Mr. Newell’s and Ms. Chung’s target performance-based cash bonus was 60% of each of their annual base salaries, respectively and Dr. Gerber’s and Mr. Chow’s targets were 40% of each of their annual base
29
salaries, respectively. The incentive target as a % of salary for each of our NEOs either remained the same in 2025 as in 2024 (Mr. Newell and Dr. Gerber), was increased as a result of promotion (Ms. Chung), or was the agreed bonus opportunity upon joining the Company (Mr. Chow) .
No amount of incentive award was guaranteed. Each NEO’s incentive award payout was based on the level of achievement of the corporate goals that were established by the Compensation Committee in early 2025, and for NEOs other than our CEO, the participant’s role in such goal achievement and the weighting of the goals.
The corporate performance goals for the fiscal year 2025 cash incentive bonus program were determined by the Compensation Committee and included certain developmental, research, financial, and operational milestones, as discussed below. These milestones were selected to drive appreciation of value in the Company, including maintaining timelines for development of the Company’s and the Company’s partners’ clinical and preclinical product candidates, successful conclusion of licensing and/or collaboration transactions, expansion and development of the Company’s manufacturing activities through third-party contract development and manufacturing organizations, maintenance of an appropriate cash position and runway, and maintaining Company culture and employee morale. Each of these goals pertain to confidential company development and business plans, the disclosure of which in any additional granularity would result in competitive harm to the company. The Compensation Committee believed that each of these goals would be challenging to achieve.
In early 2026, the Compensation Committee assessed our performance against the 2025 corporate goals, using both quantitative measurement and qualitative evaluation. The Compensation Committee determined that the Company had achieved its corporate goals at an overall level of 115% of target. In particular, the Compensation Committee assigned overachievement with respect to early pipeline progression goals, manufacturing and drug supply goals, financial performance and cultural execution goals. The 2025 corporate goals, the weighting of such goals, and the facts the Compensation Committee considered in determining the achievement of such goals are set forth below.
In assessing the 2025 corporate goals, the Compensation Committee considered achievement in the following categories with the target weightings as noted:
The Compensation Committee determined that the Company overachieved its goals with respect to Early Pipeline Progression, focusing on the progress made to bring STRO-004 to the clinic and to advance the Company’s additional preclinical programs. An IND was filed in connection with STRO-004 and clinical development was initiated nearly three months ahead of schedule. In addition, STRO-006 was nominated as a development candidate as planned and STRO-227 achieved both optimized lead and development candidate nomination in 2025; nomination of STRO-006 as development candidate in 2025 achieved a stretch goal for the Company.
With respect to the goals relating to Manufacturing and Drug Supply, the Compensation Committee determined that the Company overachieved its goals. In particular, the Compensation Committee recognized successful manufacture of STRO-004 and STRO-006 antibody intermediate in the Company’s internal manufacturing facility prior to initiation of decommissioning activities, the successful establishment of an external manufacturing network, and successful reorganization to improve development resourcing and processes following our strategic pivot in March 2025. In addition, the Compensation Committee determined that our manufacturing obligations for our collaboration partners were fully satisfied, though our agreements with our collaboration partners do not permit us to disclose the milestones achieved in these relationships because of applicable confidentiality obligations.
With regard to the Company’s financial performance, the Compensation Committee determined that the Company overachieved its goals. In particular, the Company successfully achieved its goal of ending the year with a cash and investments balance sufficient to fund activities through mid-2028, driven by the efficient winddown of luvelta
30
development and additional efficiencies resulting from the Company’s reorganizations and reductions-in-force in March and September 2025. The Compensation Committee also recognized that the Company’s financial performance in 2025 enabled a successful capital raise in early 2026.
The Compensation Committee determined that the Company overachieved its goals with respect to its commitment to execution, culture, talent and team. In particular, the Company executed an efficient strategic transition, supported employees through a difficult change management process resulting from the Company’s strategic reprioritization, and drove organizational nimbleness in support of our new early-stage priorities and programs. The Company also developed and introduced a new standard for behavioral competencies for the employees and built upon and innovated new processes, structures and standards intended to support execution, productivity, and efficiency in advancing clinical programs, scientific platform and business operations. The Company further continued development of its compliance initiatives, focusing primarily on developing policy and procedure to support the Company’s strategic pivot.
Based on our achievements during 2025, the Compensation Committee determined an overall goal achievement for the Company in 2025 of 115%. Consistent with its determination for maximum possible goal achievement in 2025, the Compensation Committee further determined that the maximum possible goal achievement for the Company in 2026 should be 150%.
NEO |
|
Target Award |
|
Actual Award |
|
Actual Award % |
||
Jane Chung, R.Ph. |
|
$ |
405,000 |
|
$ |
465,750 |
|
115% |
Hans-Peter Gerber, Ph.D. |
|
|
212,015 |
|
|
243,818 |
|
115% |
Gregory Chow |
|
|
190,000 |
|
|
218,500 |
|
115% |
William Newell |
|
|
426,600 |
|
|
— |
|
—% |
Sign-on Bonuses
From time to time, we provide one-time sign-on bonuses to newly hired executive officers as an inducement to join the Company. In 2025, Mr. Chow received a one-time cash sign-on bonus of $25,000 in connection with his appointment as Chief Financial Officer, which is fully repayable if he voluntarily terminates employment prior to the first anniversary of his start date. Sign-on bonuses are not considered part of an NEO's recurring annual compensation and are not factored into performance-based bonus target calculations.
Equity Compensation
General. We believe that equity grants provide our NEOs with the opportunity to share in increases, if any, in the value of our common stock, reinforce a long-term interest in our corporate performance, and directly motivate our NEOs to maximize long-term stockholder value. The potential realized value of certain grants depends on our stock performance and all of our equity grants utilize vesting to encourage our NEOs to continue working for us for the long term.
The Compensation Committee determines the size and type of equity awards after evaluating various factors applicable at the time of each such grant in their totality, which has included, among other things: the particular NEO’s role and responsibilities and the Compensation Committee’s view of the officer’s individual performance; the prior equity awards granted to such individual; retentive value of prior awards; our corporate performance; the value of equity grants; comparative peer data provided by its compensation consultant; and dilution to our stockholders.
All grants to executive officers require the approval of the Compensation Committee.
The 2025 grant values were meaningfully lower than the median of grant value benchmarks from the 2025 Peer Group that were reviewed by the Compensation Committee in late 2024 and early 2025. The 2025 grant values were also 34-58% lower than 2024 grant values for the two individuals who were NEOs for the full duration of both years (Ms. Chung and Dr. Gerber). Furthermore, the Compensation Committee increased the weighting of stock options
31
and decreased the weighting of RSUs in 2025 to better align the executive compensation program with our long-term performance and typical peer group practice, and stock options comprised 59-74% of our 2025 NEO grant values (rather than 50% or less in 2024).
|
|
Equity Compensation (2025 vs. 2024) |
|
|
|
|
||||||
NEO(1) |
|
2024 Equity Grant Value ($)(2)(3) |
|
|
2025 Equity Grant Value ($)(2)(4) |
|
|
2025 vs. 2024 |
|
|||
Jane Chung, R.Ph. |
|
$ |
623,360 |
|
|
$ |
412,680 |
|
|
|
-34 |
% |
Hans-Peter Gerber, Ph.D. |
|
|
407,532 |
|
|
|
171,552 |
|
|
|
-58 |
% |
Gregory Chow |
|
|
— |
|
|
|
333,938 |
|
|
N/A |
|
|
2025 Stock Options. The stock options granted to Ms. Chung and Dr. Gerber in 2025 vest equally in monthly installments over four years, while 25% of the shares subject to the stock-option granted to Mr. Chow vest 12 months after the vesting start date and the remaining shares vest equally in monthly installments over the next three years.
2025 Restricted Stock Units. The RSUs granted to our NEOs in 2025 vest equally in annual installments over four years.
Benefits Programs
Our employee benefit programs, including our 401(k) plan, employee stock purchase plan, and health and welfare programs, including health savings accounts and flexible spending arrangements, are designed to provide a competitive level of benefits to our employees generally, including our executive officers, and their families. We adjust our employee benefit programs as needed, based upon regular monitoring of applicable laws and practices and the competitive market. Our executive officers are eligible to participate in the same employee benefit plans and programs, and on the same terms and conditions, as all other U.S. full-time employees.
Perquisites and Other Personal Benefits
Currently, we do not view perquisites or other personal benefits as a significant component of our executive compensation program. Accordingly, we do not generally provide perquisites to our executive team. In 2025 and 2024, we provided a housing allowance to Ms. Chung in connection with her offer of employment; this benefit terminated in 2025. In the future, we may provide additional or different perquisites or other personal benefits in limited circumstances, such as where we believe it is appropriate to assist an individual executive in the performance of his or her duties, to make our executive team more efficient and effective and for recruitment, motivation or retention purposes. All future practices with respect to perquisites or other personal benefits will be subject to review and approval by the Compensation Committee.
Employment Agreements and Severance and Change in Control Benefits
We entered into written employment agreements with each of our NEOs that set forth the terms of their employment, including initial base salaries and eligibility to earn a bonus, as well as standard confidentiality and invention assignment agreements. Each of our named executive officers is, or in the case of Mr. Newell, was,
32
employed “at will”. These arrangements are further described under the section below titled “Executive Compensation Tables.”
Our NEOs are entitled to certain severance and change in control benefits under the terms of our executive severance and change in control plan, or Severance Plan. Below is a summary of potential post-termination compensation for our NEOs. We provide these benefits because the Compensation Committee determined that it was appropriate to provide our NEOs with severance compensation if their employment is terminated under certain circumstances. The Compensation Committee believes that the severance benefits are an important element of the NEOs’ competitive pay packages, that they serve important retention and motivation purposes and that such severance benefits, including generally requiring a release of claims against us as a condition to receiving any severance benefits, are best market practice and are in the best interest of the Company and its stockholders.
Severance Benefits
On March 17, 2021, we adopted an executive severance and change in control plan, or Severance Plan, for each of our executives with the title of vice president (or equivalent) or above, including all of our NEOs. Under the Severance Plan, if we terminate an executive other than for cause or if the executive resigns for good reason, each as defined in the Severance Plan, the executive will receive: (i) cash severance payments payable in monthly installments equal to 18 months of his or her annual base salary for our Chief Executive Officer, 15 months for our other chief executives (including the non-CEO NEOs and those chief executives with an additional title) and 9 months for our senior vice presidents and vice presidents; (ii) a pro rata portion of his or her bonus based on actual achievement of applicable metrics for the year and payable at the same time as bonuses are paid generally; (iii) payment of COBRA premiums for continued medical coverage for up to 18 months for our Chief Executive Officer, 15 months for our other chief executives and 9 months for our senior vice presidents and vice presidents; and (iv) accelerated vesting on outstanding time-based stock options and restricted stock units that vest within 18 months for our Chief Executive Officer, 15 months for our other chief executives and 9 months for our senior vice presidents and vice presidents (any performance-based equity awards will only be accelerated according to the terms of such awards).
In the event an executive’s employment terminates without cause or the executive resigns for good reason and such termination occurs on or within 18 months following a change in control, as defined in the Severance Plan, the executive will receive: (i) cash severance payments equal to 18 months of his or her annual base salary for our Chief Executive Officer, 15 months for our other chief executives and 9 months for our senior vice presidents and vice presidents, payable in a lump sum; (ii) a pro rata portion of his or her target bonus for the year, payable in a lump sum; (iii) cash severance payments equal to his or her target bonus for the year multiplied by 1.5 for our Chief Executive Officer, 1.25 for our other chief executives and 0.75 for our senior vice presidents and vice presidents, payable in a lump sum; (iv) payment of COBRA premiums for continued medical coverage for up to 18 months for our Chief Executive Officer, 15 months for our other chief executives and 9 months for our senior vice presidents and vice presidents; and (v) full accelerated vesting on all outstanding stock options and restricted stock units (any performance-based equity awards will only be accelerated according to the terms of such awards).
As a condition to receiving these severance benefits, the executive is required to execute a release of claims agreement in favor of us. The Severance Plan continues in effect for three years and shall automatically renew for successive three-year periods thereafter, unless we give notice of non-renewal prior to such renewal date. Although we have the right to amend or terminate the Severance Plan, we may not do so in any manner that diminishes any benefits being paid to an executive at the time of such amendment or termination.
Tax & Accounting Considerations
We take into account the tax effects of various forms of compensation and the potential for excise taxes to be imposed on our executive officers. There are various provisions of the Code that we consider in determining compensation, including the following:
Section 162(m). Section 162(m) of the Internal Revenue Code generally disallows a tax deduction for compensation in excess of $1 million paid to “covered employees,” which include our current NEOs. As a result, we expect that compensation awarded to our NEOs will not be deductible to the extent it is in excess of the $1 million threshold. In
33
determining the form and amount of compensation for our NEOs, the compensation committee will continue to consider all elements of the cost of such compensation, including Section 162(m). While the compensation committee considers the deductibility of awards as one factor in determining executive compensation, the compensation committee also looks at other factors in making its decisions and retains the flexibility to award compensation that it determines to be consistent with the goals of our executive compensation program even if the awards are not deductible by us for tax purposes.
Sections 280G and 4999. Any payment or benefit provided to executive officers in connection with a change-in-control transaction may be subject to an excise tax under Section 4999 of the Code. These payments also may not be eligible for a company tax deduction pursuant to Section 280G of the Code. If any of these payments or benefits are subject to the excise tax, they may be reduced to provide the individual with the best after-tax result. The individual will receive a reduced amount so that the excise tax is not triggered, or the individual will receive the full amount of the payments and benefits and then be liable for any excise tax. We have not agreed and are not otherwise obligated to provide any named executive officer with such a “gross-up” or other reimbursement.
We account for stock compensation in accordance with the authoritative guidance set forth in FASB ASC Topic 718, which requires companies to measure and recognize the compensation expense for all share-based awards made to employees and directors over the period during which the award recipient is required to perform services in exchange for the award. We determine both the grant date fair value and the service period based on applicable accounting standards. This calculation is performed for accounting purposes and reported in the compensation tables included in this proxy statement as applicable.
Additional Executive Compensation Practices, Policies and Procedures
Prohibition of Hedging and Pledging. We prohibit our NEOs (and other employees) and non-employee directors from engaging in hedging or monetization transactions involving our securities or contributing our securities to exchange funds that could be interpreted as having the effect of hedging in Company securities.
Compensation Recovery Policy
On November 12, 2023, our Board of Directors adopted a compensation recovery policy (the “Compensation Recovery Policy”) intended to comply with applicable SEC rules and Nasdaq listing standards. The Compensation Recovery Policy is administered by our Compensation Committee (in such capacity, the “Administrator”) and enables us to recover from current and former officers, and such additional employees as may be identified by the Administrator from time to time, incentive-based compensation, as defined in the Compensation Recovery Policy, in the event of an accounting restatement resulting from material noncompliance with any financial reporting requirements under federal securities laws. For more information, see the full text of our Compensation Recovery Policy, which is filed as an exhibit to our Annual Report.
Compensation Policies and Practices as they relate to Risk Management
The Compensation Committee has reviewed our executive and employee compensation programs and does not believe that our compensation policies and practices encourage undue or inappropriate risk taking or create risks that are reasonably likely to have a material adverse effect on us. The reasons for the Compensation Committee’s determination include the following:
34
Policies and Practices Related to the Grant of Certain Equity Awards
Our equity awards, including stock options, are granted in connection with our yearly compensation cycle and regularly scheduled meetings of our Compensation Committee.
During 2025, (i) none of our named executive officers were awarded stock options with an effective grant date during any period beginning four business days before the filing or furnishing of a Form 10-Q, Form 10-K, or Form 8-K that disclosed material non-public information, and ending one business day after the filing or furnishing of such reports, and (ii)
35
EXECUTIVE COMPENSATION TABLES
Summary Compensation Table
The following table presents summary information regarding the total compensation for services rendered in all capacities that was earned by our named executive officers during the years ended December 31, 2025 and 2024, as applicable.
Name and Principal Position |
|
Year |
|
Salary($) |
|
|
Bonus($)(4) |
|
|
Option Awards($)(1) |
|
|
Stock Awards($)(1) |
|
|
Non-Equity Incentive Plan Compensation |
|
|
All Other Compensation |
|
|
Total($) |
|
|||||||
Jane Chung, R.Ph. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
Chief Executive Officer |
|
2025 |
|
|
652,000 |
|
|
|
— |
|
|
|
245,280 |
|
|
|
167,400 |
|
|
|
465,750 |
|
|
|
45,500 |
|
(3) |
|
1,575,930 |
|
|
|
2024 |
|
|
560,000 |
|
|
|
— |
|
|
|
169,360 |
|
|
|
454,000 |
|
|
|
182,000 |
|
|
|
78,000 |
|
(3) |
|
1,443,360 |
|
William J. Newell |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
Former Chief Executive Officer |
|
2025 |
|
|
239,693 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,431,618 |
|
(5) |
|
1,671,311 |
|
|
|
2024 |
|
|
707,500 |
|
|
|
— |
|
|
|
589,373 |
|
|
|
758,180 |
|
|
|
277,290 |
|
|
|
— |
|
|
|
2,332,343 |
|
Hans-Peter Gerber, Ph.D. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
Chief Scientific Officer |
|
2025 |
|
|
527,532 |
|
|
|
— |
|
|
|
110,127 |
|
|
|
61,425 |
|
|
|
243,818 |
|
|
|
— |
|
|
|
942,902 |
|
|
|
2024 |
|
|
512,500 |
|
|
|
100,000 |
|
|
|
203,232 |
|
|
|
204,300 |
|
|
|
133,900 |
|
|
|
— |
|
|
|
1,153,932 |
|
Gregory Chow(6) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
Chief Financial Officer |
|
2025 |
|
|
277,083 |
|
|
|
25,000 |
|
|
|
245,938 |
|
|
|
88,000 |
|
|
|
218,500 |
|
|
|
— |
|
|
|
854,522 |
|
Offer Letters with NEOs
We have entered into written employment agreements with each of our NEOs that set forth the terms of their employment, including initial base salaries and eligibility to earn a bonus, as well as standard confidentiality and invention assignment agreements. Each of our named executive officers is employed “at will.” The offer letters for Mr. Newell also included certain severance protections, which severance protections have been superseded by the
36
Severance Plan described above in the section entitled “-Employment Agreement and Severance and Change in Control Benefits -Severance Benefits.”
Mr. Newell’s Employment Separation and Transition Agreement
On March 13, 2025, we and Mr. Newell entered into a Separation Agreement (the “Newell Separation and Transition Agreement”) in connection with a Board approved leadership transition; the Board treated Mr. Newell’s departure as a termination without cause under the Severance Plan and entered into the Newell Separation and Transition Agreement. Pursuant to the Newell Separation and Transition Agreement, Mr. Newell ceased his role as the Company’s President and CEO and resigned as a director of the Board, effective March 13, 2025. In addition, pursuant to the Newell Separation and Transition Agreement, Mr. Newell continued to provide transition services to the Company through May 1, 2025.
In connection with Mr. Newell’s execution of a general release and waiver of claims and the terms of the Newell Separation and Transition Agreement, and pursuant to the terms of the Severance Plan, Mr. Newell received the following benefits: (i) two cash payments totaling the gross amount of $1,066,500.00, equal to 150% of his annual salary, with the first payment paid in 2025 and the second payment paid on January 15, 2026; (ii) eighteen months of accelerated vesting on all equity awards under our 2018 Equity Incentive Plan, (iii) a lump sum payment reflecting Mr. Newell’s bonus opportunity for the 2025 fiscal year on a pro-rated basis, based on our actual percentage achievement of our 2025 corporate goals; (iv) and coverage under the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended paid for by us for up to 18 months beginning June 1, 2025. In addition, the post-termination option exercise period of Mr. Newell’s outstanding and vested options with an exercise price of less than $120 was extended for 2 years.
Ms. Chung’s Employment Offer Letter and Amended Offer Letter
Effective as of the Newell Separation Date, the Board appointed Ms. Chung to succeed Mr. Newell as Chief Executive Officer. In connection with Ms. Chung’s appointment as Chief Executive Officer, we entered into an amended offer letter with Ms. Chung (the “Amended Chung Offer Letter”). Pursuant to the Amended Chung Offer Letter, Ms. Chung (i) received a base salary of $675,000, (ii) was eligible to earn a target bonus of 60% of her base salary and (iii) was granted an award of 62,000 stock options and an award of 31,000 restricted stock units, both awards to be granted at the time of our annual 2025 refresh equity grants. Ms. Chung also continued to be a participant in the Severance Plan, now as a Tier 1 Participant as defined in the Severance Plan, described above in
the section entitled “-Employment Agreement and Severance and Change in Control Benefits -Severance Benefits”.
Dr. Gerber’s Employment Offer Letter
Dr. Gerber is party to an offer letter with us dated August 28, 2023, pursuant to which he serves as our Chief Scientific Officer. The terms and conditions of his offer letter provide for an annual base salary, and eligibility for an annual bonus, a sign-on bonus of $100,000, which would have been fully repayable had Dr. Gerber chosen to leave our company prior to the second anniversary of his employment, health insurance and other benefits. Dr. Gerber is an at-will employee. Dr. Gerber participates in the Severance Plan described above in the section entitled “-Employment Agreement and Severance and Change in Control Benefits -Severance Benefits”.
Mr. Chow’s Employment Offer Letter
Mr. Chow is party to an offer letter with us dated May 22, 2025, pursuant to which he serves as our Chief Financial Officer. The terms and conditions of his offer letter provide for an annual base salary, and eligibility for an annual bonus, a sign-on bonus of $25,000 which will be fully repayable if Mr. Chow chooses to leave the company prior to the first anniversary of his employment, health insurance and other benefits. Mr. Chow is an at-will employee. Mr. Chow participates in the Severance Plan described above in the section entitled “-Employment Agreement and Severance and Change in Control Benefits -Severance Benefits”.
37
2025 Outstanding Equity Awards at Fiscal Year-End Table
The following table presents, for each of our named executive officers, information regarding outstanding stock options and restricted stock units held as of December 31, 2025.
|
|
Option Awards |
|
Stock Awards |
|
|||||||||||||||||||||
Name |
|
Grant Date |
|
Number of Securities Underlying Unexercised Options (#) Exercisable |
|
|
Number of Securities Underlying Unexercised Options (#) Unexercisable |
|
|
Option Exercise Price ($) |
|
|
Option Expiration Date |
|
Grant Date |
|
Number of Units of Stock That Have Not Vested (#) |
|
|
Market Value of Units of Stock That Have Not Vested ($) |
|
|||||
William J. Newell |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
3/5/2024 |
(1)(2) |
|
11,595 |
|
|
|
— |
|
|
|
45.40 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
3/3/2023 |
(1)(2) |
|
262 |
|
|
|
— |
|
|
|
57.90 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
3/3/2023 |
(1)(2) |
|
11,194 |
|
|
|
— |
|
|
|
57.90 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
3/4/2022 |
(1)(2) |
|
1,037 |
|
|
|
— |
|
|
|
81.70 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
3/4/2022 |
(1)(2) |
|
15,562 |
|
|
|
— |
|
|
|
81.70 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
1/29/2020 |
(1)(2) |
|
291 |
|
|
|
— |
|
|
|
100.90 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
1/29/2020 |
(1)(2) |
|
12,208 |
|
|
|
— |
|
|
|
100.90 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
1/29/2019 |
(1)(2) |
|
927 |
|
|
|
— |
|
|
|
104.50 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
|
|
1/29/2019 |
(1)(2) |
|
43,572 |
|
|
|
— |
|
|
|
104.50 |
|
|
5/1/2027 |
(6) |
|
|
|
|
|
|
|
||
Jane Chung, R.Ph. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
4/4/2025 |
(1)(2) |
|
— |
|
|
|
20,247 |
|
|
|
5.40 |
|
|
4/5/2035 |
|
|
|
|
|
|
|
|
||
|
|
4/4/2025 |
(1)(2) |
|
11,624 |
|
|
|
30,128 |
|
|
|
5.40 |
|
|
4/5/2035 |
|
|
|
|
|
|
|
|
||
|
|
3/5/2024 |
(1)(2) |
|
— |
|
|
|
1,562 |
|
|
|
45.40 |
|
|
3/5/2034 |
|
|
|
|
|
|
|
|
||
|
|
3/5/2024 |
(1)(2) |
|
2,184 |
|
|
|
1,253 |
|
|
|
45.40 |
|
|
3/5/2034 |
|
|
|
|
|
|
|
|
||
|
|
12/1/2023 |
(1)(2) |
|
— |
|
|
|
1,626 |
|
|
|
27.50 |
|
|
12/1/2033 |
|
|
|
|
|
|
|
|
||
|
|
12/1/2023 |
(1)(2) |
|
1,748 |
|
|
|
126 |
|
|
|
27.50 |
|
|
12/1/2033 |
|
|
|
|
|
|
|
|
||
|
|
3/3/2023 |
(1)(2) |
|
732 |
|
|
|
1,254 |
|
|
|
57.90 |
|
|
3/3/2033 |
|
|
|
|
|
|
|
|
||
|
|
3/3/2023 |
(1)(2) |
|
2,013 |
|
|
|
— |
|
|
|
57.90 |
|
|
3/3/2033 |
|
|
|
|
|
|
|
|
||
|
|
3/4/2022 |
(1)(2) |
|
3,937 |
|
|
|
263 |
|
|
|
81.70 |
|
|
3/4/2032 |
|
|
|
|
|
|
|
|
||
|
|
8/9/2021 |
(4)(5) |
|
16,000 |
|
|
|
— |
|
|
|
183.70 |
|
|
8/9/2031 |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4/4/2025 |
(1)(3) |
|
31,000 |
|
|
|
358,670 |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3/5/2024 |
(1)(3) |
|
7,500 |
|
|
|
86,775 |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
12/1/2023 |
(1)(3) |
|
5,000 |
|
|
|
57,850 |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3/3/2023 |
(1)(3) |
|
1,376 |
|
|
|
15,920 |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3/4/2022 |
(1)(3) |
|
788 |
|
|
|
9,117 |
|
|||
Hans-Peter Gerber |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
12/23/2025 |
(1)(2) |
|
— |
|
|
|
6,922 |
|
|
10.38 |
|
|
12/23/2035 |
|
|
|
|
|
|
|
|
|||
|
|
12/23/2025 |
(1)(2) |
|
— |
|
|
|
1,003 |
|
|
10.38 |
|
|
12/23/2035 |
|
|
|
|
|
|
|
|
|||
|
|
4/4/2025 |
(1)(2) |
|
2,296 |
|
|
|
9,954 |
|
|
|
5.40 |
|
|
4/4/2035 |
|
|
|
|
|
|
|
|
||
|
|
3/5/2024 |
(1)(2) |
|
2,625 |
|
|
|
3,375 |
|
|
|
45.40 |
|
|
3/4/2034 |
|
|
|
|
|
|
|
|
||
|
|
9/18/2023 |
(4)(5) |
|
9,843 |
|
|
|
7,657 |
|
|
|
37.70 |
|
|
9/18/2033 |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4/4/2025 |
(1)(3) |
|
11,375 |
|
|
|
131,609 |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3/5/2024 |
(1)(3) |
|
3,375 |
|
|
|
39,049 |
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
9/18/2023 |
(3)(4) |
|
7,500 |
|
|
|
86,775 |
|
|||
Gregory Chow |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
12/23/2025 |
(1)(2) |
|
— |
|
|
|
8,200 |
|
|
|
10.38 |
|
|
12/23/2035 |
|
|
|
|
|
|
|
|
||
|
|
6/16/2025 |
(4)(5) |
|
— |
|
|
|
27,500 |
|
|
|
8.80 |
|
|
6/16/2035 |
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
6/16/2025 |
(3)(4) |
|
10,000 |
|
|
|
115,700 |
|
|||
38
39
PAY VERSUS PERFORMANCE
In accordance with rules adopted by the Securities and Exchange Commission pursuant to Section 953(a) of the Dodd-Frank Wall Street Reform and Consumer Protection Act of 2010, we provide the following disclosure regarding executive compensation for our principal executive officer (“PEO”) and our other NEOs (“Other NEOs”) and Company performance for the fiscal years listed below. The Compensation Committee did not consider the pay versus performance disclosure below in making its pay decisions for any of the years shown. The amounts shown for Compensation Actually Paid have been calculated in accordance with Item 402(v) of Regulation S-K and do not reflect compensation actually earned, realized, or received by the Company’s NEOs; these amounts reflect the Summary Compensation Table total with certain adjustments as described in the following table and footnotes.
|
|
SCT Total Comp. for PEO: Ms. Chung(1) |
|
|
Compensation Actually Paid to PEO: Ms. Chung(2) |
|
|
SCT Total Comp. for Prior PEO: Mr. Newell(3) |
|
|
Compensation Actually Paid to Prior PEO: Mr. Newell(2) |
|
|
Average Summary Compensation Paid to Other NEOs(4) |
|
|
Average Compensation Actually Paid to Other NEOs(2)(3) |
|
|
Value of Initial $100 Investment Based On Sutro TSR(5) |
|
|
Net Income ($M) |
|
||||||||
2025 |
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
( |
) |
|||||||
2024 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
( |
) |
|||||||
2023 |
|
|
|
|
|
|
|
|
|
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
( |
) |
||||||
2025 - Greg Chow, Hanspeter Gerber; 2024 - Edward Albini, Jane Chung; 2023 - Anne Borgman, Hanspeter Gerber
40
|
|
CEO |
|
|||||
|
|
2025 - Chung |
|
|
2025 - Newell |
|
||
|
|
|
|
|
|
|
||
Summary Compensation Table Total |
|
$ |
|
|
$ |
|
||
Grant Date Fair Value of Stock and Option Awards in the Covered Year |
|
|
- |
|
|
|
|
|
Fair Value at Year-End of Unvested Stock and Option Awards Granted in the Covered Year* |
|
|
+ |
|
|
|
|
|
Fair Value of Stock and Option Awards Granted in the Covered Year that Vested in the Covered Year* |
|
|
+ |
|
|
|
|
|
Change in Fair Value of Unvested Stock and Option Awards Granted in Prior Years* |
|
|
- |
|
|
|
- |
|
Change in Fair Value of Stock and Option Awards from Prior Years that Vested in the Covered Year* |
|
|
- |
|
|
|
- |
|
Change in Fair Value of Stock and Option Awards from Prior Years that were Forfeited in the Covered Year* |
|
|
|
|
|
- |
|
|
Compensation Actually Paid |
= |
$ |
|
|
$ |
|
||
|
|
Average of Other NEOs(4) |
|
|
|
|
2025 |
|
|
|
|
|
|
|
Summary Compensation Table Total |
|
$ |
|
|
Grant Date Fair Value of Stock and Option Awards in the Covered Year |
|
|
- |
|
Fair Value at Year-End of Unvested Stock and Option Awards Granted in the Covered Year* |
|
|
+ |
|
Fair Value of Stock and Option Awards Granted in the Covered Year that Vested in the Covered Year* |
|
|
+ |
|
Change in Fair Value of Unvested Stock and Option Awards Granted in Prior Years* |
|
|
- |
|
Change in Fair Value of Stock and Option Awards from Prior Years that Vested in the Covered Year* |
|
|
- |
|
Compensation Actually Paid |
= |
$ |
|
|
* All stock option valuations included in "Compensation Actually Paid" values were performed using the Black-Scholes option pricing model in a manner generally consistent with the process used to determine stock option grant date fair values under FASB ASC Topic 718 (refer to our annual report for additional detail). All stock award valuations included in "Compensation Actually Paid" values were performed using closing price of the Company's common stock.
41
The relationship between Compensation Actually Paid (CAP) and the financial performance elements reflected in the above Pay versus Performance tables are described in the below charts:
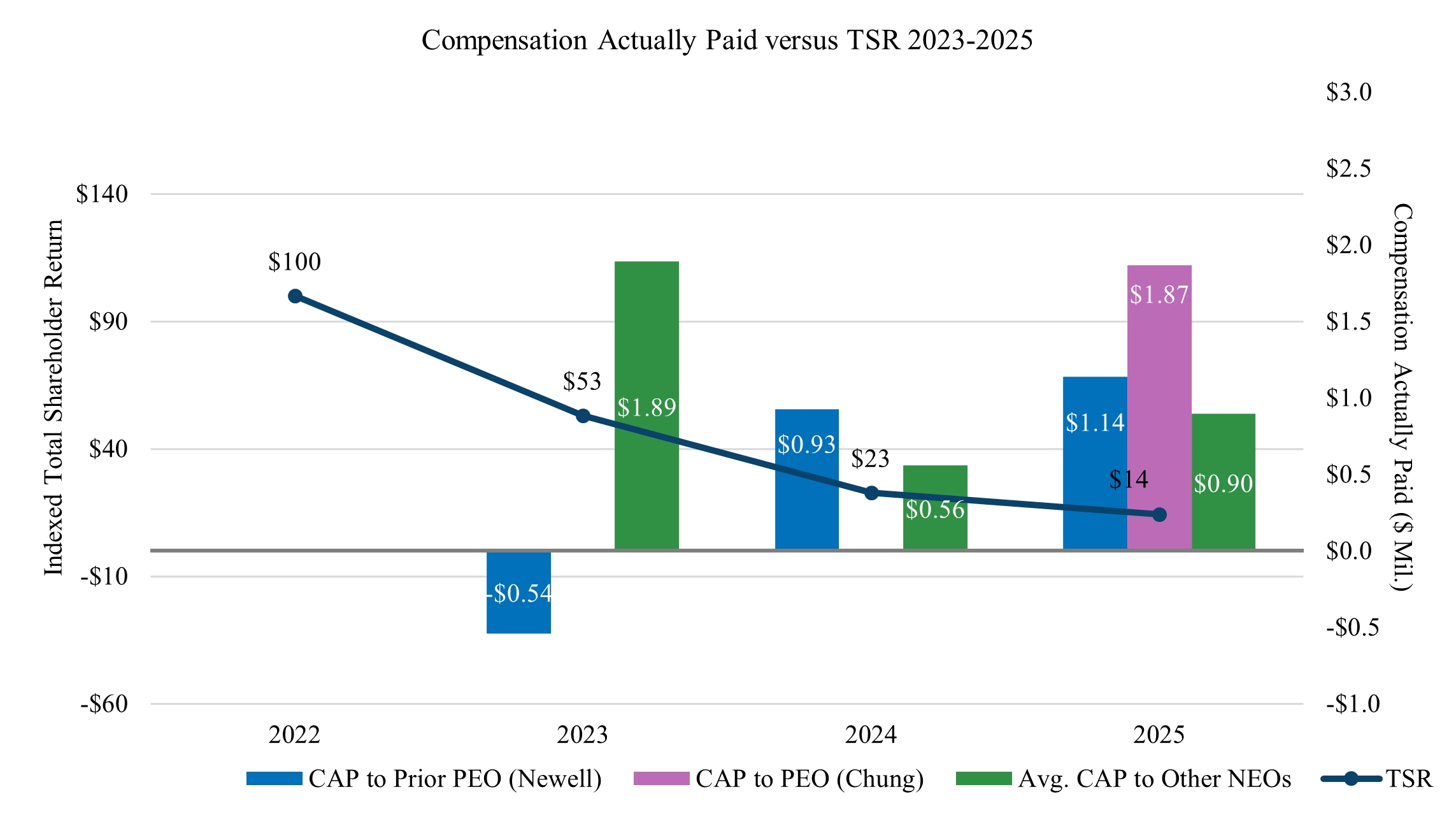
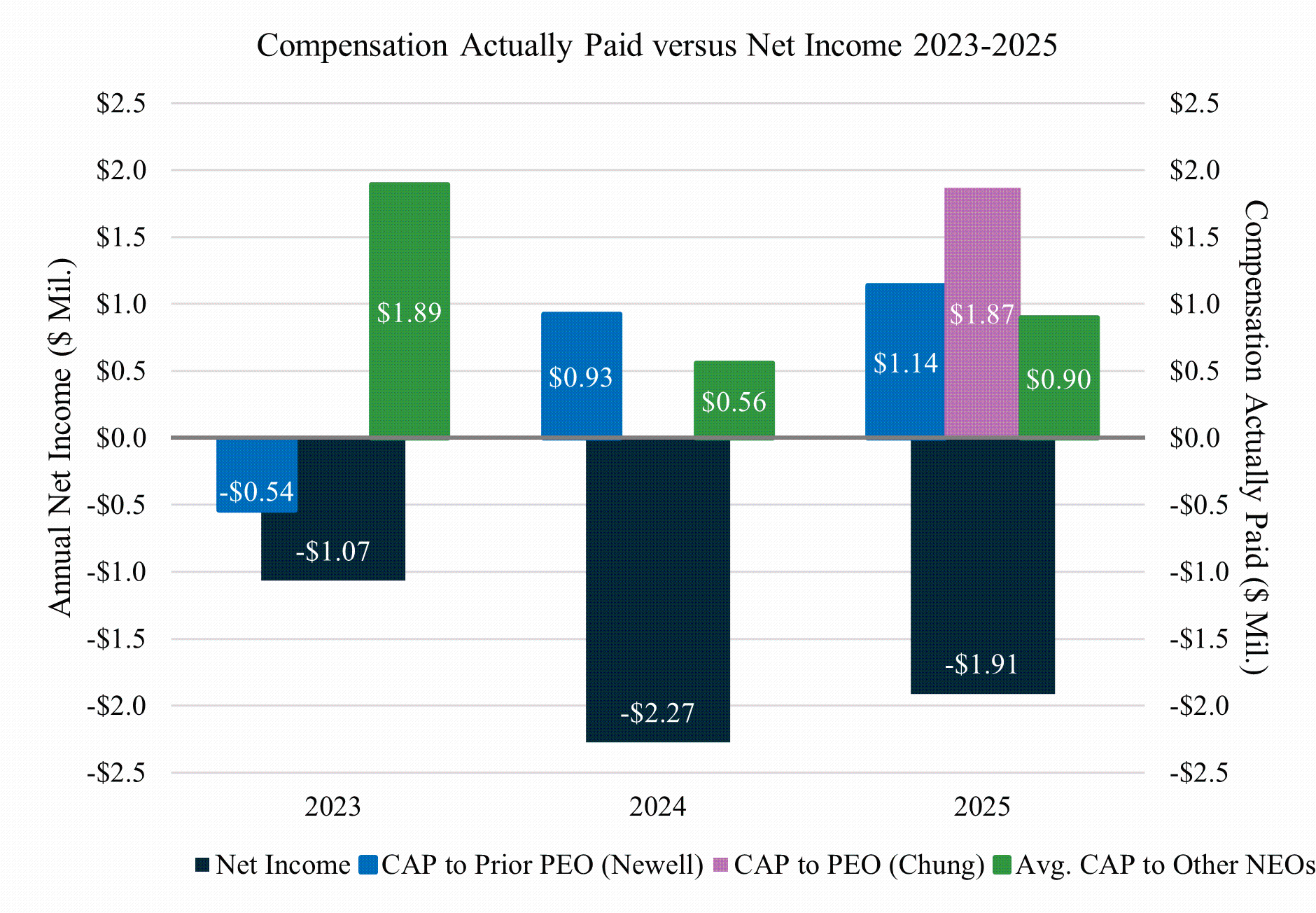
42
EQUITY COMPENSATION PLAN INFORMATION
The following table presents information as of December 31, 2025 with respect to compensation plans under which shares of our common stock may be issued.
Plan category |
|
Number of securities to be issued upon exercise of outstanding options, warrants and rights |
|
|
Weighted-average exercise price of outstanding options, warrants and rights (1) |
|
|
Number of securities remaining available for future issuance under equity compensation plans (2) |
|
Equity compensation plans approved by security holders |
|
1,213,687 |
(3) |
|
$ 55.86 |
|
|
501,882 |
|
Equity compensation plans not approved by security holders |
|
134,653 |
(4) |
|
55.87 |
|
|
244,006 |
(5) |
Total |
|
1,348,340 |
|
|
$ 55.86 |
|
|
745,888 |
|
43
PROPOSAL NO. 3
ADVISORY VOTE ON EXECUTIVE COMPENSATION
In accordance with Section 14A of the Exchange Act, we are including in this proxy statement the opportunity for our stockholders to vote to approve, on a non-binding, advisory basis, the compensation of our named executive officers as disclosed in this proxy statement. This non-binding advisory vote is commonly referred to as a “say on pay” vote. We are requesting that stockholders vote, in an advisory capacity, on our named executive officer compensation as disclosed in the “Executive Compensation” section of this proxy statement at the Annual Meeting.
We strongly encourage stockholders to review the information contained in the “Executive Compensation-Compensation Disclosure” section of this proxy statement for additional details on our compensation of our Named Executive Officers, including our compensation philosophy and objectives, as well as the processes the Compensation Committee used to determine the structure and amounts of the compensation of our Named Executive Officers. The Compensation Committee and the Board believe that these policies and procedures are effective in implementing our compensation philosophy and in achieving its goals. We are asking you to indicate your support for the compensation of our Named Executive Officers as described in this Proxy Statement. This vote is not intended to address any specific item of compensation, but rather the overall compensation of our Named Executive Officers and the philosophy, policies and practices described in this Proxy Statement.
RESOLVED, that our stockholders approve, on a non-binding advisory basis, the compensation of the named executive officers, as disclosed in the proxy statement pursuant to Item 402 of Regulation S-K, including the compensation tables and narrative discussion and the other related disclosures.”
While the results of this advisory vote are not binding, the Compensation Committee will consider the outcome of the vote in deciding whether to take any action as a result of the vote and when making future compensation decisions for our named executive officers.
OUR BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” APPROVAL OF PROPOSAL NO. 3
44
From January 1, 2024 to the present, there have been no transactions, and there are currently no proposed transactions, in which the amount involved exceeds the lesser of $120,000 and 1.0% of the average of our total assets during the last two completed fiscal years to which we or any of our subsidiaries was (or is to be) a party and in which any director, director nominee, executive officer, holder of more than 5% of our capital stock, or any immediate family member of or person sharing the household with any of these individuals, had (or will have) a direct or indirect material interest, except for payments set forth under “Proposal No. 1 Election of Class II Directors” and “Executive Compensation” above.
Policies and Procedures for Related-Person Transactions
Our Board of Directors has adopted a written related-person transactions policy. Under this policy, our executive officers, directors, nominees for election as a director, beneficial owners of more than 5% of our common stock, and any members of the immediate family of and any entity affiliated with any of the foregoing persons, are not permitted to enter into a material related-person transaction with us without the review and approval of our Audit Committee, or a Committee composed solely of independent directors in the event it is inappropriate for our Audit Committee to review such transaction due to a conflict of interest. The policy provides that any request for us to enter into a transaction with an executive officer, director, nominee for election as a director, beneficial owner of more than 5% of our common stock or with any of their immediate family members or affiliates in which the amount involved exceeds $120,000 will be presented to our Audit Committee for review, consideration and approval. In approving or rejecting any such proposal, our Audit Committee will consider the relevant facts and circumstances available and deemed relevant to the Audit Committee, including, but not limited to, whether the transaction is on terms no less favorable than terms generally available to an unaffiliated third party under the same or similar circumstances and the extent of the related person’s interest in the transaction.
ADDITIONAL INFORMATION
Stockholder Proposals to be Presented at Next Annual Meeting
Requirements for Stockholder Proposals to be Brought Before an Annual Meeting. Our Amended and Restated Bylaws provide that for stockholder nominations to our Board of Directors or other proposals to be considered at an annual meeting of shareholders, the stockholder must give timely notice thereof in writing to the Corporate Secretary at Sutro Biopharma, Inc., 111 Oyster Point Boulevard, South San Francisco, California, 94080.
To be timely for our company’s annual meeting of shareholders to be held in 2027 (2027 Annual Meeting), a stockholder’s notice must be delivered to or mailed and received by our Corporate Secretary at our principal executive offices not earlier than 5:00 p.m. Eastern Time on February 5, 2027, and not later than 5:00 p.m. Eastern Time on March 7, 2027. A stockholder’s notice to the Corporate Secretary must set forth as to each matter the stockholder proposes to bring before the 2027 Annual Meeting the information required by applicable law and our Amended and Restated Bylaws. However, if the date of the 2027 Annual Meeting is more than 30 days before or more than 70 days after the one-year anniversary of the date of our 2026 Annual Meeting, for the stockholder notice to be timely, it must be delivered to the Corporate Secretary at our principal executive offices not earlier than the close of business on the 120th day prior to the currently proposed annual meeting and not later than the close of business on the later of (1) the 90th day prior to such annual meeting or (2) the close of business on the 10th day following the day on which public announcement of the date of such meeting is first made by us. To comply with our Amended and Restated Bylaws as well as the universal proxy rules, stockholders who intend to solicit proxies in support of director nominees other than our nominees for 2027 Annual Meeting must ensure that our Corporate Secretary receives written notice that sets forth all information required by our Amended and Restated Bylaws and by Rule 14a-19(b) under the Exchange Act within the time frames set forth above.
Requirements for Stockholder Proposals to be Considered for Inclusion in our Proxy Materials. Stockholder proposals submitted pursuant to Rule 14a-8 under the Exchange Act and intended to be presented at our 2027 Annual Meeting must be received by us not later than December 23, 2026, in order to be considered for inclusion in our proxy materials for that meeting. A stockholder’s notice to the Corporate Secretary must set forth as to each
45
matter the stockholder proposes to bring before the 2027 Annual Meeting the information required by applicable law and our Amended and Restated Bylaws.
Available Information
The Annual Report on Form 10-K is also available at https://ir.sutrobio.com/.
“Householding” - Stockholders Sharing the Same Address
The SEC has adopted rules that permit companies and intermediaries (such as brokers) to implement a delivery procedure called “householding.” Under this procedure, multiple stockholders who reside at the same address may receive a single copy of our Annual Report on Form 10-K and proxy materials, including the Notice of Internet Availability, unless the affected stockholder has provided other instructions. This procedure reduces printing costs and postage fees, and helps protect the environment as well.
We expect that a number of brokers with account holders who are our stockholders will be “householding” our Annual Report on Form 10-K and proxy materials, including the Notice of Internet Availability. A single Notice of Internet Availability and, if applicable, a single set of our Annual Report on Form 10-K and other proxy materials will be delivered to multiple stockholders sharing an address unless contrary instructions have been received from one or more of the affected stockholders. Once you have received notice from your broker that it will be “householding” communications to your address, “householding” will continue until you are notified otherwise or until you revoke your consent. Stockholders may revoke their consent at any time by contacting their broker. Stockholders may revoke their consent at any time by contacting Equiniti Trust Company LLC, either by calling toll-free (800) 937-5449, or by writing to Equiniti Trust Company LLC Operations Center, 6201 15th Avenue, Brooklyn, New York 11219.
Upon written or oral request, we will undertake to promptly deliver a separate copy of the Notice of Internet Availability and, if applicable, Annual Report on Form 10-K and other proxy materials to any stockholder at a shared address to which a single copy of any of those documents was delivered. To receive a separate copy of the Notice of Internet Availability and, if applicable, Annual Report on Form 10-K and other proxy materials, you may write our Investor Relations Department at Sutro Biopharma, Inc., 111 Oyster Point Boulevard, South San Francisco, California, 94080, Attn: Investor Relations, or at IR@sutrobio.com.
Any stockholders who share the same address and currently receive multiple copies of our Notice of Internet Availability or Annual Report on Form 10-K and other proxy materials who wish to receive only one copy in the future can contact their bank, broker or other holder of record to request information about “householding” or our Investor Relations Department at the address or telephone number listed above.
Delinquent Section 16(a) Reports
Section 16 of the Exchange Act requires our directors, certain officers, and beneficial owners of more than ten percent of our common stock to file reports with the SEC indicating their holdings of and transactions in the company’s equity securities. Based solely on a review of such copies and written representations from our reporting persons, we believe that all Section 16(a) filing requirements were timely met during 2025, except that a late Form 4 was filed for Jane Chung on August 26, 2025 reporting the vesting and settlement of RSUs and the related net withholding of shares by us to satisfy tax withholding and remittance obligations on August 9, 2025 and a late Form 4 was filed for Barbara Leyman on August 26, 2025, reporting the vesting and settlement of RSUs and the related net withholding of shares by us to satisfy tax withholding and remittance obligations on July 8, 2025.
OTHER MATTERS
Our Board of Directors does not presently intend to bring any other business before the meeting and, so far as is known to the Board of Directors, no matters are to be brought before the meeting except as specified in the notice of the meeting. As to any business that may arise and properly come before the meeting, however, it is intended that
46
proxies, in the form enclosed, will be voted in respect thereof in accordance with the judgment of the persons voting such proxies.
47

Sutro biopharma logo SUTRO BIOPHARMA, INC. 1 1 1 OYSTER POINT BOULEVARD SOUTH SAN FRANCISCO, CALIFORNIA 94080 Your Vote Counts! SUTRO BIOPHARMA, INC.2026 Annual Meeting Vote by June 04, 2026 11:59 PM ET You invested in SUTRO BIOPHARMA, INC. and it's time to vote! You have the right to vote on proposals being presented at the Annual Meeting. This is an important notice regarding the availability of proxy material for the shareholder meeting to be held on June 05, 2026. Get informed before you vote View the Notice & Proxy Statement, Form 10-K online OR you can receive a free paper or email copy of the material(s) by requesting prior to May 22, 2026. If you would like to request a copy of the material(s) for this and/or future shareholder meetings, you may (1) visit www.ProxyVote.com, (2) call 1-800-579-1639 or (3) send an email to sendmaterial@proxyvote.com. If sending an email, please include your control number (indicated below) in the subject line. Unless requested, you will not otherwise receive a paper or email copy. For complete information and to vote, visit www.ProxyVote.com Control # Smartphone users Point your camera here and vote without entering a control number Virtually at the Meeting* June 05, 2026 8:00 AM PDT virtually at: www.virtualshareholdermeeting.com/STR02026*Please check the meeting materials for any special requirements for meeting attendance.

Vote at www.ProxyVote.comTHIS IS NOT A VOTABLE BALLOT This is an overview of the proposals being presented at the upcoming shareholder meeting. Please follow the instructions on the reverse side to vote these important matters. Voting Items Board Recommends 1. Election of Class Il Directors Nominees: 01 ) Jane Chung, RPh 02) Connie Matsui 03) James Panek For 2. To ratify the appointment of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026. 3. To approve, on a non-binding, advisory basis, the compensation of our named executive officers. For For NOTE: Such other business as may properly come before the meeting or any adjournment thereof. Prefer to receive an email instead? While voting on www.ProxyVote.com, be sure to click "Delivery Settings".

SUTRO BIOPHARMA, INC.111 OYSTER POINT BOULEVARD SOUTH SAN FRANCISCO, CALIFORNIA 94080 SCAN TO VIEW MATERIALS & VOTE VOTE BY INTERNET - www.proxyvote.com or scan the QR Barcode above Use the Internet to transmit your voting instructions and for electronic delivery of information. Vote by 11:59 P.M. ET on 06/04/2026. Have your proxy card in hand when
you access the web site and follow the instructions to obtain your records and to create an electronic voting instruction form.During The Meeting - Go to www.virtualshareholdermeeting.com/STRO2026 You may attend the meeting via the Internet and vote during the meeting. Have the information that is printed in the box marked by the arrow available and follow the instructions.VOTE BY PHONE - 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions. Vote by 11:59 P.M. ET on 06/04/2026. Have your proxy card in hand when you call and then follow the instructions.VOTE BY MAIL Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717 TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: KEEP THIS PORTION FOR YOUR RECORDS THIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED DETACH AND RETURN THIS PORTION ONLY The Board of Directors recommends you vote FOR the following: For Withhold For All All All Except To withhold authority to vote for any individual nominee(s), mark “For All Except” and write the number(s) of the nominee(s) on the line below.Election of Class II Directors Nominees 01) Jane Chung, RPh 02) Connie Matsui 03) James Panek The Board of Directors recommends you vote FOR proposals 2 and 3. For Against Abstain
2. To ratify the appointment of Ernst & Young LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026.3. To approve, on a non-binding, advisory basis, the compensation of our named executive officers. NOTE: Such other business as may properly come before the meeting or any adjournment thereof.Please sign exactly as your name(s) appear(s) hereon. When signing as attorney, executor, administrator, or other fiduciary, please give full title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or partnership name by authorized officer.Signature [PLEASE SIGN WITHIN BOX] Date Signature (Joint Owners) Date

Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting: The Notice and Proxy Statement and Form 10-K are available at www.proxyvote.com SUTRO BIOPHARMA, INC.Annual Meeting of Shareholders June 5, 2026 8:00 AM PDT This proxy is solicited by the Board of Directors The undersigned hereby appoints Jane Chung, R.Ph and David Pauling, and each of them, with full power of substitution and power to act alone, as proxies to vote all the shares of Common Stock which the undersigned would be entitled to vote if personally present and acting at the Annual Meeting of Shareholders of SUTRO BIOPHARMA, INC., to be held on June 5, 2026, via a live webcast at www.virtualshareholdermeeting.com/STRO2026, and at any adjournments or postponements thereof. This proxy, when properly executed, will be voted in the manner directed herein. If no such direction is made, this proxy will be voted FOR ALL NOMINEES in Proposal 1, FOR Proposal 2 and FOR Proposal 3.Continued and to be signed on reverse side
